Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:

A material widely used in electronics and other fields turns out to have a split personality. Researchers from the Weizmann Institute of Science have shown that the surface of strontium titanate differs from the bulk of this material in a major, previously unknown way. So significant is this difference that Nature magazine has commented in its News & Views section: “A crystal’s surface has been found to behave as a distinct material.”
Strontium titanate crystals are so brilliant they were once used to make artificial gems, but currently they are best known as substrates for growing transistors, oxide-based thin films and various nanostructures. The Weizmann researchers have reported in Advanced Materials that although these crystals are nonpolar, a nanolayer on their surface is pyroelectric – that is, it generates electricity when heated. This means that this nanolayer is polar: It has positive and negative electric poles.

Understanding this distinction within crystals is crucial for the design of various devices because electric polarity can critically affect a material’s properties. The Weizmann finding must therefore be taken into account when building devices containing strontium titanate, and it may also have broader implications, suggesting that polarity may exist on the surface of other inorganic crystals as well.
“Surface polarity in an inorganic material is a new concept,” says team leader Prof. Igor Lubomirsky of the Materials and Interfaces Department. “No one had thought of looking for it because it seems counterintuitive.”
Several years ago, Lubomirsky and departmental colleague Prof. Meir Lahav had shown that temperature-dependent polarity can emerge on the surface of nonpolar organic crystals, such as the amino acids glycine, L-alanine and DL-serine. But inorganic crystals are more rigidly symmetrical than organic ones, so they appear to be unlikely candidates for surface polarity, which by definition requires that this symmetry be broken, for example by distortions in the material’s structure. Still, Lubomirsky and Lahav realized that symmetry is broken in inorganic crystals as well – on their surface – and this led them to hypothesize that just maybe polarity can, against all odds, be present in nonpolar inorganic crystals.
No one had thought of looking for it because it seems counterintuitive
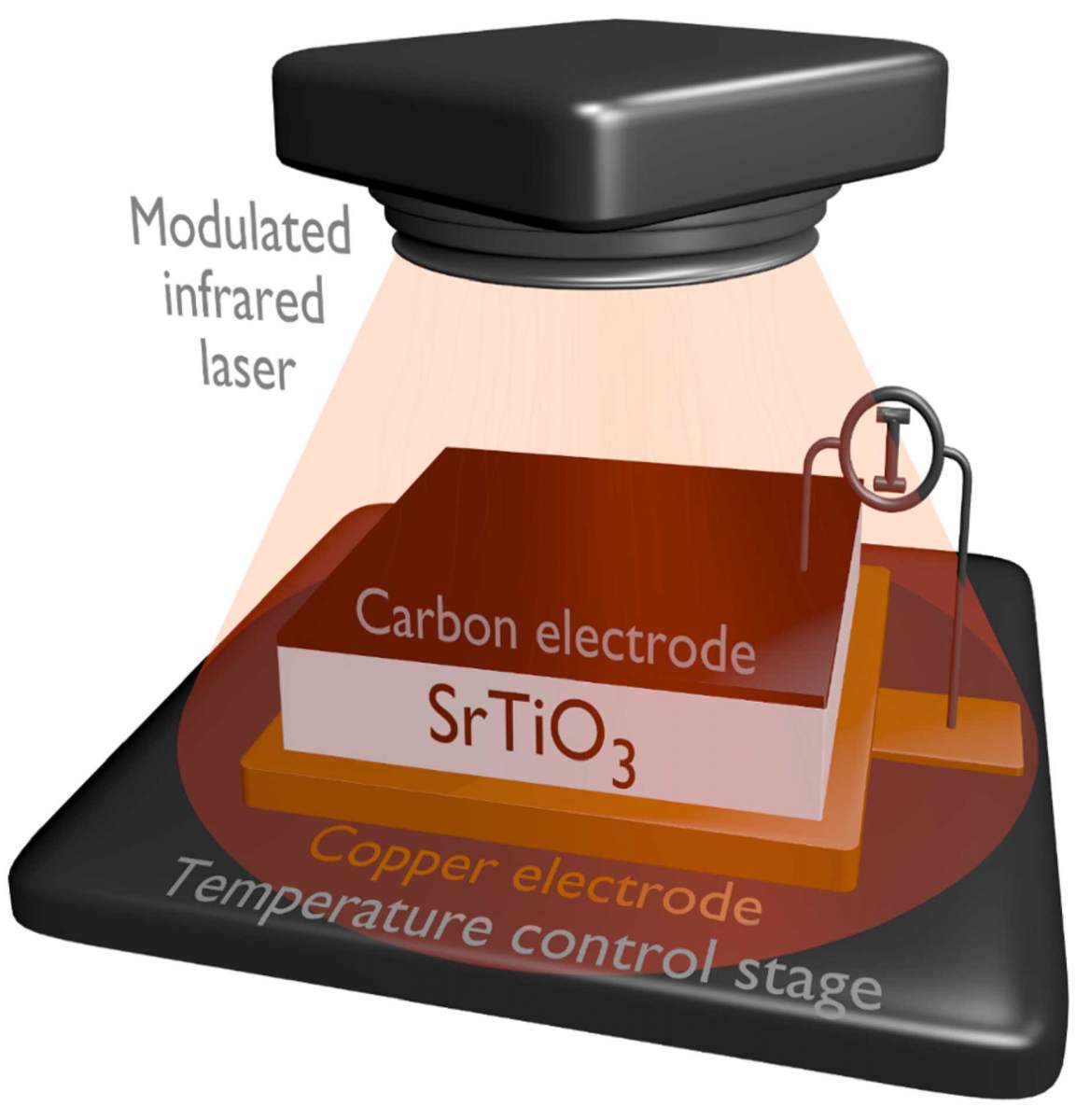
To confirm this hypothesis, the researchers would have to prove that any pyroelectricity discovered in an inorganic crystal comes from the crystal’s surface rather than its bulk – a task that presented an enormous technological challenge. Dr. Elena Meirzadeh, then a doctoral student, and other team members made use of a special installation built in Lubomirsky’s lab, which makes it possible to measure extremely small electric currents, of about 10-12, or 0.000000000001 amperes – that is, about a ten billion times less than electric current generated by a standard AA battery – at time intervals of several microseconds (millionths of a second). The researchers heated the surface of strontium titanate crystals with a laser beam of rapidly changing intensity and measured the electric current generated by the heating. They found that such current was indeed produced, but it decayed at great speed, which suggested that it was present only in the outermost layer – one just several atoms thick, measuring about one nanometer.
The researchers performed additional experiments to rule out the possibility that the current originated in the bulk of the material rather than its surface. In one of these they deposited an ultrathin layer of non-crystalline silica on top of the strontium titanate and found that the pyroelectricity disappeared, adding support to the notion that it had been a surface effect.
After conducting a theoretical analysis of their findings, Lubomirsky and colleagues concluded that pyroelectricity had emerged due to a common surface distortion known as a “surface relaxation.” Because such relaxation occurs in many crystals, the researchers have suggested that surface pyroelectricity might be found in a wide range of materials. They have also proposed that the effect they discovered can help design multilayered devices in which the polarity of the various layers will be exploited to produce desired properties, leading to new applications in electronics, nanotechnology, and energy harvesting.
The Weizmann discovery further suggests that the surface of strontium titanate and similar inorganic crystals may hold additional surprises. In particular, Nature’s News & Views commentary suggests that although strontium titanate is an insulator, its surface may turn out to be one of the much sought-after polar metals, exotic materials expected to have unique electronic properties. According to the commentary, the Weizmann findings “hint that polar metals might have been under our noses all along, paradoxically on the surfaces of non-polar insulators.”
Study participants included Evgeniy Makagon and Dr. David Ehre of Weizmann’s Materials and Interfaces Department; Drs. Hagai Cohen and Irit Rosenhek-Goldian of Weizmann’s Chemical Research Support Department; Dr. Dennis V. Christensen, Dr. Arghya Bhowmik, Dr. Juan Maria G. Lastra and Prof. Nini Pryds of the Technical University of Denmark; Prof. Erie H. Morales of Seton Hall University; and Prof. Andrew M. Rappe of the University of Pennsylvania.
Prof. Igor Lubomirsky's research is supported by the Angel Faivovich Foundation for Ecological Research; the Ullmann Family Foundation; the Jacob and Charlotte Lehrman Foundation; the Harold Perlman Family;and the estate of Olga Klein – Astrachan. Prof. Lubomirsky is the incombent of the Rowland and Sylvia Schaefer Professorial Chair in Energy Research.