Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
Expanding wave fronts – ripples on a lake, for example – are a well-studied physical phenomenon. These sorts of dynamics can also be seen in biological systems, in which information is often passed from cell to cell. Prof. Roy Bar-Ziv and research students Alexandra Tayar and Eyal Karzbrun of the Weizmann Institute’s Materials and Interfaces Department, in collaboration with Prof. Vincent Noireaux from the University of Minnesota, created an experimental wave front of spreading information through cells, and showed that, like setting dominoes to fall in a row, fixing the initial conditions in these cells created a self-propagating yet highly controlled expansion.

The “cell-on-a-chip” system that the scientists used is one that Bar-Ziv and his group have labored on for over a decade. These cells, around a millionth of a meter in depth, are etched in silicon, and a dense forest of DNA strands is attached to the bottom of each cell. A number of cells are then connected by thin capillary tubes to a larger channel. Once the researchers fill the artificial cells with the genes of their choice, they add an extract of E. coli that contains everything they need (including the ribosomes) to translate the genetic information to proteins. These proteins tend to move out of the cell and, through the capillary tubes, into the larger channel, mimicking the way that living biological cells manage their protein turnover.
In the new experiment, which was reported in Nature Physics, the scientists added more thin capillaries, this time hooking the cells up serially. One capillary connected the first cell in line to the second, the next tube connected the second to the third, and so on. They then inserted genes encoding certain regulatory proteins – proteins that turn on genes – into the cells. When the protein was produced in the first cell, it passed into the second, activating genes and causing the protein to be produced in the next cell down the line, on to the end of the line. These proteins also activated genes for reporter proteins that glow in fluorescent colors, so the researchers were able to track their concentration in each cell. Indeed, the researchers noted a progressive “ripple” passing from cell to cell as each began producing proteins. This “wave of light” passed down the line of cells unhindered and untouched by human hands. “What we see in this system is a domino effect,” says Tayar. “And the system is self-perpetuating. It is based on protein communication – one to regulate, another which acts as a switch; together they keeps the system going. As gene translation is kindled in cell after cell, we see the reporter proteins light up in a series.”
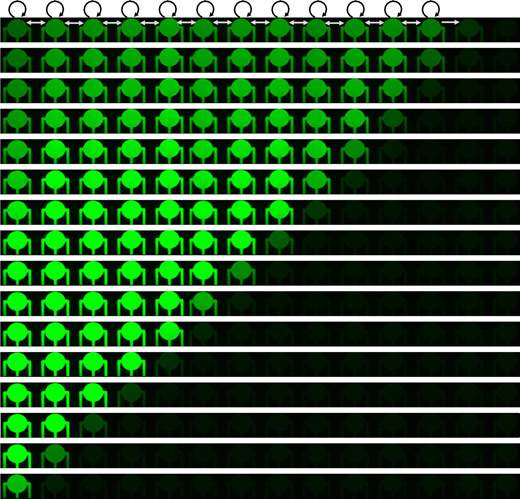
“This phenomenon – a wave of genetic information – is similar to the way information is transferred in an optic communications cable,” says Karzbrun. “The self-propagating wave retains its form from beginning to end. By precisely fixing the amounts of the different genes, we were able to control many aspects of this propagation, including the rate and the size of the ripples.”
Bar-Ziv points out that this type of cell-to-cell information transfer is involved in such dynamic biological processes as early embryonic development. “In the future,” he says, “this system may be used to recreate and investigate such biological processes as spatial patterning or the dynamics of genetic cycles. Systems of artificial cells on chips may also be used for sensing, communicating and processing information, and the monitoring of various environmental phenomena.”
Prof. Roy Bar Ziv's research is supported by the Yeda-Sela Center for Basic Research; the Gurwin Family Fund for Scientific Research; and the estate of Raymond Lapon.