At the end of the 1970s, Yonath was a young researcher at the Weizmann Institute with an ambitious plan to settle one of the major outstanding questions concerning living cells: She wanted to determine the three-dimesional structure of the ribosome – the cell’s factory for translating into proteins the instructions written in the gene code – and thus to reveal the mechanics guiding the process. This was the beginning of a long quest – one that would take decades and in which she would be met with reactions of disbelief and even ridicule in the scientific community. In her Nobel lecture, Yonath compared her journey to climbing Mt. Everest “only to discover that a higher Everest stood in front of us.” She began her work in collaboration with Prof. H. G. Wittmann of the Max Planck Institute for Molecular Genetics in Berlin, who supported her studies academically and financially, in parallel to maintaining her modest lab at the Weizmann Institute with an even more modest budget. Yonath came, over the years, to lead a large team of researchers from all corners of the globe. Though her research began as an attempt to understand one of the fundamental components of life, it has led to a detailed understanding of the actions of some of the most widely prescribed antibiotics. Her findings may not only aid in the development of more efficient antibacterial drugs, they could give scientists new weapons in the fight against antibiotic-resistant bacteria – a problem that has been called one of the most pressing medical challenges of the 21st century
Almost all the functions of every living organism are performed by proteins, which carry out the daily processes of life. Each protein is made up of a sequence of building blocks called amino acids, and their sequence, which is encoded in the genes, determines how that protein will fold up into the three-dimensional structure it needs to carry out its assigned task. The ribosome is a sort of machine that can make any protein, fashioned according to the code fed into it. The machinery is composed of two subunits – one large and one small – that exist independently but come together to form a functional complex specifically for the task of producing proteins.
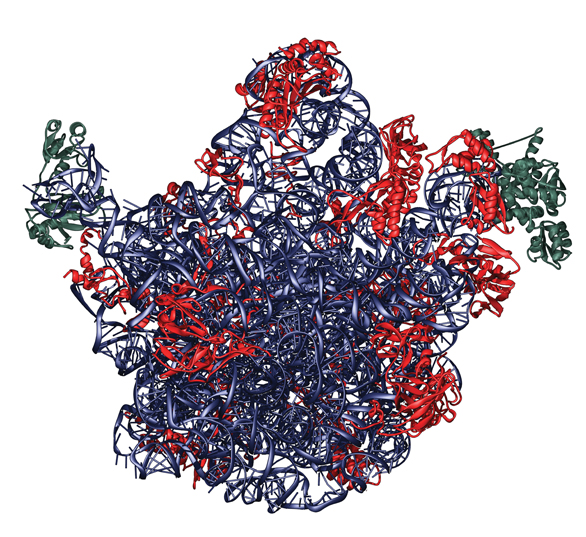
Because the ribosome is so central to life (not to mention interesting), scientists around the world had been trying for many years to figure out how it works; but without an understanding of its spatial structure, there was little hope of forming a comprehensive picture of its actions. To reveal the three-dimensional structure of such small entities as biological molecules – too small to be seen even under an electron microscope – scientists first turn them into crystals and then bombard the crystals with powerful X rays. The shapes of the crystallized compounds are revealed in the patterns of radiation scattered from them, each unique pattern resulting from the compound’s internal chemical composition. With such X-ray crystallography techniques, one can “map” the distribution of the electrons in the investigated molecule and deduce its structure. But when dealing with ribosomes, there are additional challenges: The ribosome is a complex of proteins and RNA; its structure is extraordinarily intricate; it’s unusually flexible; it’s unstable; and it lacks internal symmetry, a feature that makes the crystallization of even the subunits an extremely formidable task.
Making crystals
At the beginning of the 1980s, Yonath – working in both the Weizmann Institute in Israel and the Max Planck Institute in Germany – created the first ribosome crystals in the world. The procedure, which she developed especially for this purpose, included a method for the preparation of the crystallizable ribosome that had been developed at the Weizmann Institute by Profs. Ada Zamir, Ruth Miskin, Nahum Sonenberg, David Elson and Meir Wilchek. Her inspiration, she says, came from an article on polar bears: “Polar bears pack their ribosomes in an orderly way in their cells just before hibernation, and these stay intact and functional for months. I said: ‘If polar bears know how to do it, we can do it too.’” She was able to produce the first ribosome crystals in a fairly short time. The early images she managed to obtain, however, yielded only the fuzziest picture, and were not sufficiently detailed to explain the workings of the ribosome.
Yonath was also the first to visualize the tunnel running through the active ribosome, through which the nascent protein progresses as it’s being formed – until it’s big enough to take on a three-dimensional structure and “look after itself.” In the course of her research she developed a number of new techniques that are today widely used in structural biology labs around the world. One of these is cryo bio-crystallography, which involves exposing the crystal to extremely low temperatures – -185° Celsius – to prevent the structure’s disintegration under the X-ray bombardment. She also developed a unique experimental system based on ribosomes taken from the hardy bacteria living in the extreme environments of the Dead Sea, thermal springs and atomic piles.
By the end of the 1990s, Yonath had succeeded in breaking the resolution barrier, thanks to improvements both in the crystals and in the facilities for detecting the X-ray diffraction. The result was the first “electron density map” of the ribosome’s small subunit. This is the piece of the ribosome that does the actual job of decoding the gene code, which it reads from a strand of messenger RNA. Her findings were published in 1999 in the Proceedings of the National Academy of Sciences (PNAS), USA. Then, in 2000 and 2001, Yonath published the first complete three-dimensional structure of both subunits of a bacterial ribosome – work that was hailed by Science magazine as one of the ten most important breakthroughs of the year.
These discoveries were clearly a high point in 20 years of research. But Yonath’s quest to understand the ribosome was far from complete. Armed with new insight into ribosomal structure, she moved on to revealing what that structure can tell us about its actions and, especially, how antibiotic drugs block those actions in bacterial ribosomes. Another challenge Yonath and her research team undertook was to try to understand how cellular factors initiate the process, “telling” the ribosome that it must begin protein assembly. To produce an image of the contacts with one such cellular factor, Yonath and her colleagues inserted a component into the crystal that attaches to the ribosome and activates it.
Drug effects
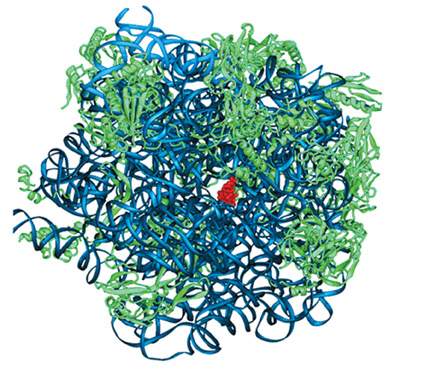
Because ribosomes are so essential to life, it makes sense that many antibiotic drugs work by targeting their actions. “The advances we made in our long quest to solve the structure and function of the ribosome may pave the way toward better antibiotic drugs,” says Yonath. Together with team members Dr. Anat Bashan, research student Raz Zarivach and scientists at the Max Planck Institute in Germany, she crystallized bacterial ribosomes, each with one of five antibiotic compounds bound to it. They found that the drugs tended to bind in specific “pockets” in the structure, preventing the ribosomes from manufacturing proteins and eventually killing the bacteria. Since these findings were published in Nature, in 2001, Yonath has revealed the mode of action of several more common antibiotics, and her research in this area is ongoing. Yonath: “Most antibiotic drugs bind to one active site or another in the ribosomes, paralyzing them and preventing them from making the proteins necessary for bacterial life. With our understanding of protein manufacture, we can now design antibiotics to attack bacteria that have developed resistance to the existing drugs.”
Another line of research – conducted in collaboration with Profs. Lou Massa of City University of New York and Nobel laureate (1985, Physics) Jerome Karle of the Naval Research Laboratory in Washington – used a method called “quantum crystallography” to observe the process of protein assembly in real time.
Planning her future research, Yonath intends to look to the distant past. Ribosomes are found in every living being – from yeast and bacteria to mammals – and the structure of their active sites has been extraordinarily well-preserved throughout evolution. Could a pared-down, primordial ribosome have begun producing the first proteins hundreds of millions of years ago, essentially giving rise to life? How did these first ribosomes come into being? How did they begin to produce proteins? How did they evolve into the sophisticated protein factories we see today in living cells? Yonath plans on answering these and other questions in her future work.
Prof. Ada Yonath’s research is supported by the Helen and Milton A. Kimmelman Center for Biomolecular Structure and Assembly. Prof. Yonath is the Martin S. and Helen Kimmel Professor of Structural Biology.
Prof. Yonath was asked to deliver the Nobel banquet speech in the name of the three Chemistry laureates. The following is the text of that speech:
Your Majesties, Your Royal Highnesses, Ladies and Gentlemen,
It is a great honor for me to be able to express my sincere gratitude to the Nobel Foundation.
I was asked to represent the 2009 Chemistry laureates, who are three different people. We belong to different nationalities, genders, views and upbringings. However, we share a common passion: to understand how the ribosome makes proteins according to the instructions of the genetic code, an issue to which many more scientists around the world have contributed tremendously.
When I was approached by the Royal Academy, I was advised to benefit from the speech given by Isaac Bashevis Singer, the 1978 Literature laureate, who started with: “People ask me often, ‘Why do you write in a dying language, Yiddish?’”
Indeed, words originating from the verb “to die” were frequently used when I described my initial plans to determine the ribosome structure. Many distinguished scientists said: “Why work on ribosomes, they are dead… we know all what can be known about them,” or: “This is a dead end road,” or: “You will be dead before you get there.” To my satisfaction, these predictions were proven wrong, the ribosomes are alive and kicking (so am I) and their high-resolution structures stimulated more advanced studies as well as the imagination of many youngsters, including my granddaughter, Noa, who is showing continuous interest, and invited me at the ages of 5 and 13 to explain to her classes what the ribosome is. Also, there is a new saying in Israel: Curly hair (like mine) means ROSH MALEH RIBOSOMIM, which translates to: head full of ribosomes. Furthermore, our studies added to the buzz around the lovely polar bears, which inspired my own research and are now endangered by the changing climate.
I was also instructed not to use this opportunity for thanking. I have always been known as an obedient person, and thus decided to follow this advice too, but with one exception. I want to thank warmly my dedicated chauffeur, Nisse. Without him I would have been lost in Stockholm, the wonderful, albeit dark city, and consequently miss most of the fascinating events of this magic week.


First Experiment

As a young child, Prof. Ada Yonath lived in a four-room apartment that her family shared with another three families and their children. Hard conditions didn’t dampen her enormous curiosity, though. Already at five, she was actively investigating the world around her. She tried to measure the height of their tiny balcony using furniture from inside the apartment. She put a table on another table and then a chair and a stool on top, and when she climbed up on her structure, she fell and broke her arm. In this photo from her kindergarten class, Ada (standing, center) has her arm in a cast.
















