We all begin life as a collection of embryonic stem cells that have a remarkable ability to mature into any cell type in the body. Even in adulthood, our bodies harbor small populations of adult stem cells that can develop into various mature cell types and regenerate certain tissues. Stem cells are in no hurry to mature and, like Peter Pan, can remain youthful for many years. Now, researchers from the Weizmann Institute of Science and the Hebrew University of Jerusalem reveal that these perpetual adolescents of the cellular world sometimes forgo longevity, mature into potent antimicrobial cells and, when infected by bacteria, die in an act of cellular altruism – thereby protecting the tissue from persistent infection. The findings, published in Nature Immunology, uncover an active role that stem cells play in immune defense and suggest that this remodeling of stem cells may be implicated in inflammatory diseases and cancer.

All the cells in our body carry the same genetic material, but different cells express different sets of genes. When stem cells mature – a process known as differentiation – they switch certain genes on and others off, gradually implementing the genetic program of the mature cell they are becoming and acquiring its characteristics. Maturity, however, comes at a steep cost: The lifespan of a stem cell can shrink from many years to just weeks or even days.
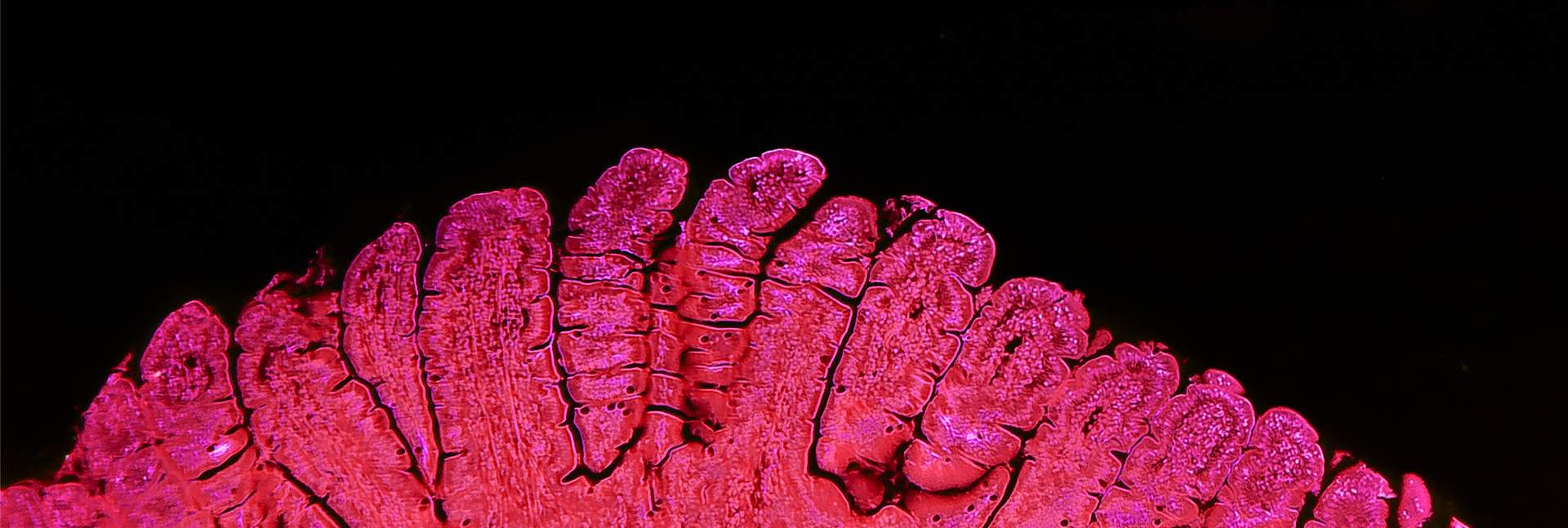
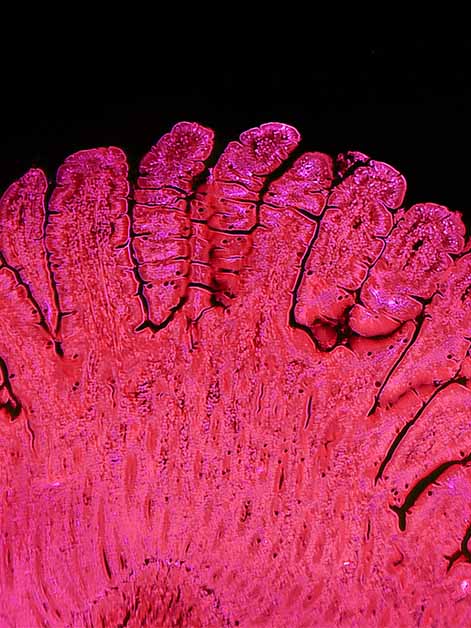
In the new study – led by Dr. Sacha Lebon in Dr. Moshe Biton’s lab at the Weizmann Institute in collaboration with Dr. Matan Hofree of the Hebrew University – the researchers focused on the inner lining, or epithelium, of the small intestine, which is characterized by rapid and continuous cell turnover. These cells perform a wide range of functions, from absorbing nutrients and secreting mucus to taking part in immune defense.
""In effect, an infected stem cell ‘sacrifices’ itself, accepting a shortened lifespan in order to prevent prolonged infection"
In experiments in mice, the scientists examined what happens to this tissue following infection with Salmonella. Within a day, the bacteria had invaded a substantial fraction of the stem cells responsible for tissue renewal. Yet instead of causing widespread destruction, cell turnover actually accelerated. Single-cell genomic sequencing, which reveals the active genetic programs in individual cells, showed that infected stem cells were more likely to undergo enhanced maturation, differentiating into specific epithelial cell types with antibacterial functions.
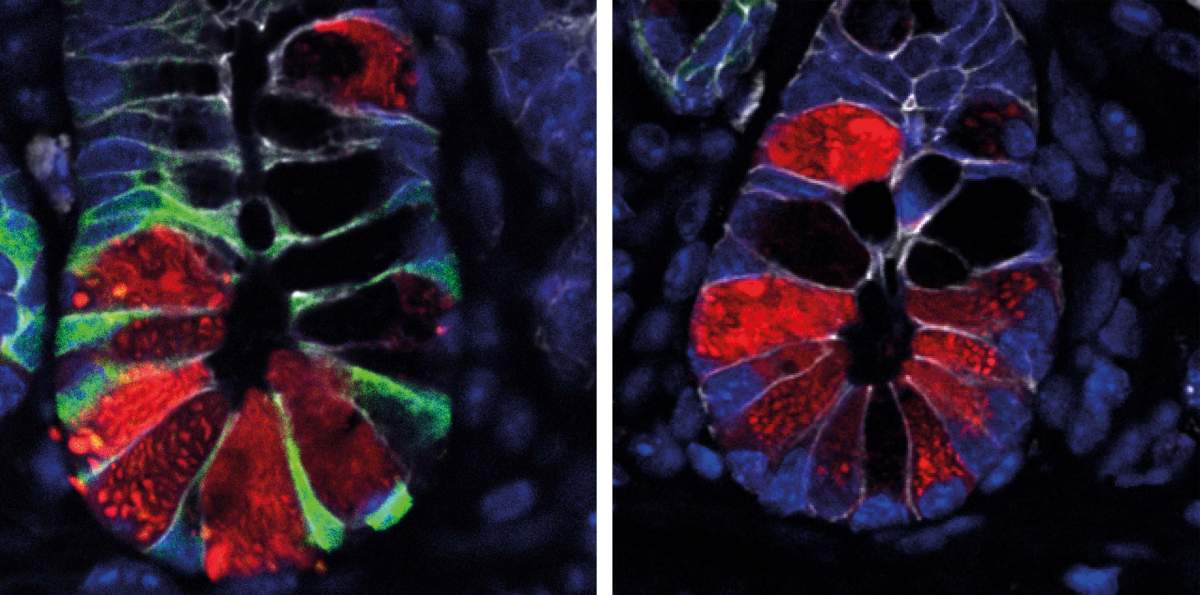
“We found that intestinal stem cells enhance their maturation when invaded by bacteria,” says Biton. “Because these cells normally divide every day, maturation ensures that infected cells do not continue to proliferate. In effect, an infected stem cell ‘sacrifices’ itself, accepting a shortened lifespan in order to prevent prolonged infection and allow the remaining stem cells to restore the tissue. Another advantage of this mechanism is that stem cells differentiate in a targeted manner into epithelial cells that produce antibacterial substances, thereby bolstering the immune defenses.”
Until now, it was widely believed that stem cells, despite their importance, are not capable of defending themselves and instead rely on the protection provided by surrounding immune cells. However, maturation in response to infection has now been revealed as an intrinsic defense mechanism of stem cells – one that proved effective even in genetically engineered mice lacking a functional immune system. The researchers found that each intestinal stem cell contains protein complexes, known as inflammasomes, that act as “smoke detectors” for bacterial invasion. While similar detectors exist in innate immune cells, only in stem cells do they trigger an accelerated maturation program in response to infection.

The scientists discovered that this newly identified immune mechanism also operates in lab-grown human intestinal models (organoids) infected with Salmonella. In collaboration with Prof. Yael Haberman Ziv of Sheba Medical Center, they identified a link between activation of this mechanism and specific bacterial species commonly found in people with Crohn’s disease.
“Inflammation is the body’s response to harmful or foreign agents – it helps tissues heal but can become destructive when it spirals out of control and turns chronic,” explains Biton. “We hypothesize that excessive differentiation of stem cells into mature cells with antibacterial activity may contribute to the exaggerated, chronic inflammation seen in Crohn’s disease. Alternatively, accelerated maturation may be a consequence of the disease, since Crohn’s is associated with frequent bacterial infections, and stem cells may be working overtime to cope. In any case, it is now clear that studying stem cells is essential for understanding inflammatory diseases in humans.”

In the future, accelerated stem cell maturation may prove to be an effective defense mechanism not only against bacterial invasion but also against other forms of damage. It is possible that cellular “smoke detectors” can also sense genetic damage, inflammation or metabolic disruption, triggering differentiation to halt the division of damaged cells. In this way, the mechanism may shorten the lifespan of compromised cells and help prevent disease. “I hope these discoveries will ultimately contribute to improved treatments for inflammatory diseases and cancer,” says Biton.