Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
Despite extensive research, scientists are still unsure about the causes of autism spectrum disorders (ASDs) – a broad range of conditions characterized by impaired cognition, difficulties with social interaction and communication, and typical repetitive behaviors. Severe maternal infection during pregnancy has been linked to a higher prevalence of ASD, yet this link remains controversial.
Weizmann Institute of Science’s Prof. Orly Reiner and PhD student Lihi Ben-Reuven of the Molecular Genetics Department thought that mimicking viral infections during pregnancy in mice could help determine how these affect brain development in the embryo. “Although signs of autism usually only first appear around age two or three years, it is considered a neurodevelopmental disease, which suggests that changes could already be ‘brewing’ in the embryonic brain,” says Reiner.
Using a substance that mimics a virus thus activated the pregnant mouse's immune response. When the activation was carried out on day 12 (out of 18 days of pregnancy in mice), the offspring exhibited autism-like behaviors, and the researchers found that their cortex (the frontal part of the brain) was thicker than usual. The cortex is the part of the brain where most information processing occurs – including thinking, perceiving, and producing and understanding language. These are also the processes affected in those with ASD.

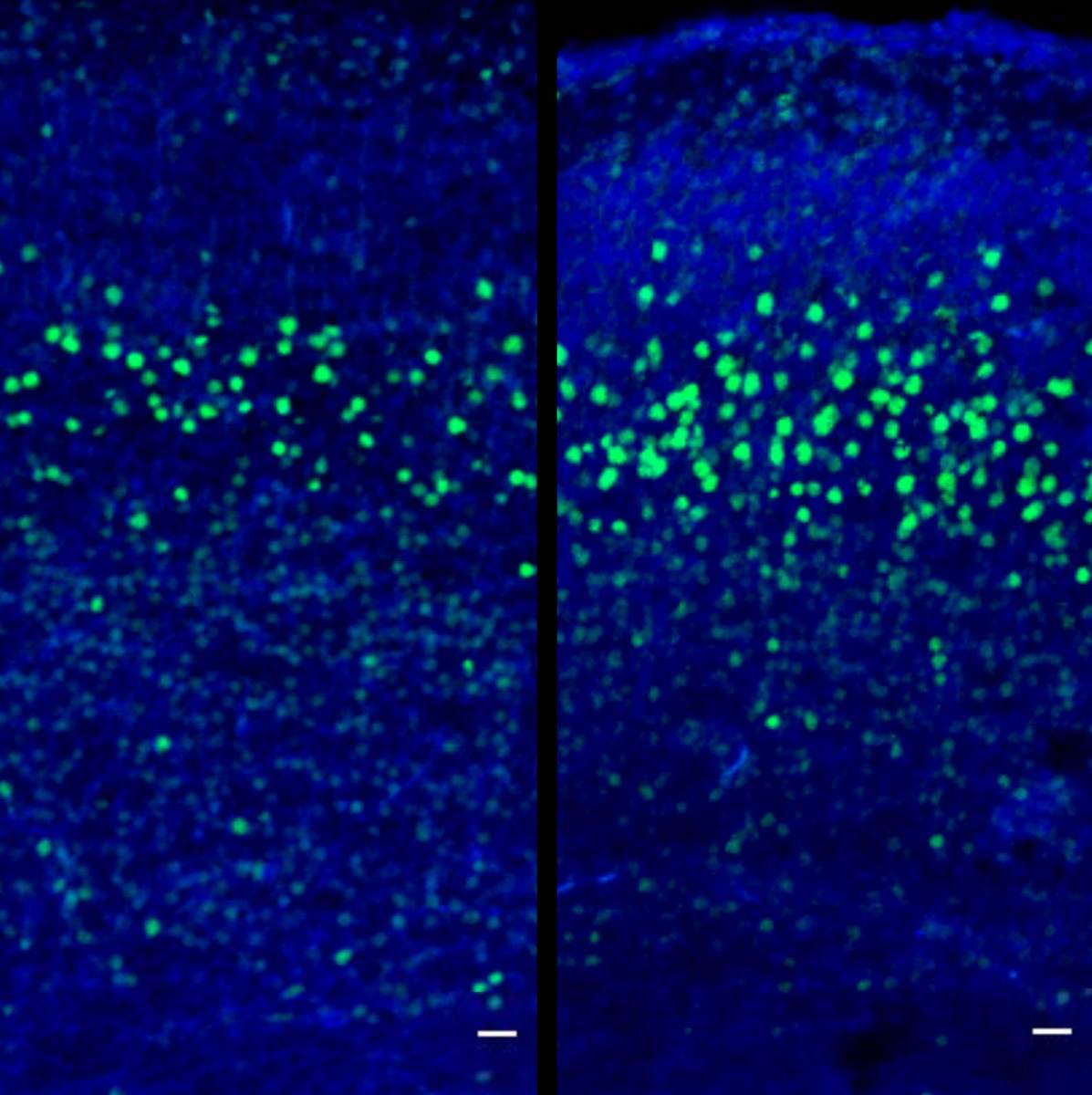
Taking their study a step further, Reiner and Ben-Reuven investigated what was happening in the embryos’ cortex immediately following activation of the immune response. Surprisingly, they found that more neurons than usual were being “born” as little as six hours following the immune activation. “In other words, the mother got sick and the embryos were affected,” explains Ben-Reuven. The researchers discovered that the production of additional neurons was limited to a 48-hour time window.
But which neurons were being overproduced? And how? Previous studies had demonstrated that following activation of the mothers’ immune response, there is an increase in the levels of a signaling molecule called interleukin 6 (IL6). IL6 is known to be linked to inflammation caused by viruses. But it can also increase the “birth rate” of brain cells. In the current study, the scientists noticed that the increased levels of IL6 right away shortened the cell cycles of the neuron-producing stem cells in the embryos' cortex. As a result, more neurons were generated in this part of embryonic brain.
As the embryonic brain develops, the cortex organizes itself into six layers. Each layer is composed of particular types of neurons, and these have complicated patterns of connections. The production of the different types of neurons is guided by transcription factors – proteins that help to turn specific genes “on” or “off.” The researchers found increased levels of a particular transcription factor, which in turn resulted in an overproduction of a specific type of neuron. These neurons are the CTIP2+ neurons that are found in layer five of the cortex. “As a result, at the end of pregnancy and in the young mice, the offspring have a relatively larger cortex and a thickened layer five,” explains Ben-Reuven. "These offspring displayed abnormal behavior; and we could now tie this behavior to the acute immune activation during a particular developmental stage. Our ability to discover the overproduction of specific types of neurons in an autism model is really interesting.”
This research was recently published in Molecular Psychiatry. Overall, it lends support to the notion that an acute inflammatory immune response ‒ during a specific time window in pregnancy ‒ does, indeed, affect embryonic brain development. With regard to humans, Reiner cautions: “Increased brain size has been found in a subgroup of autism patients. Whether or not there is a direct correlation, time will tell, but if a link is proven in the future, we are talking about a very short and a very severe infection – not something like the common cold.”
Prof. Orly Reiner is Head of the M. Judith Ruth Institute for Preclinical Brain Research. Her research is also supported by the Helen and Martin Kimmel Institute for Stem Cell Research; the Nella and Leon Benoziyo Center for Neurological Diseases; the David and Fela Shapell Family Center for Genetic Disorders Research; the Brenden-Mann Women’s Innovation Impact Fund; the Richard F. Goodman Yale/Weizmann Exchange Program; the Irving B. Harris Fund for New Directions in Brain Research; the Irving Bieber, M.D. and Toby Bieber, M.D. Memorial Research Fund; the Leff Family; Barbara and Roberto Kaminitz; Sergio and Sônia Lozinsky; Debbie Koren; Jack and Lenore Lowenthal; and the Dears Foundation. Prof. Reiner is the incumbent of the Bernstein-Mason Chair of Neurochemistry.