Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
“Bad cholesterol” is a misnomer: Cholesterol is a basic component of life. Specifically, it provides elasticity to the fatty substance that makes up the membranes of our cells. Even the LDL cholesterol, which is known to be a risk factor in artery disease, is not really “bad” all on its own. LDL is the name given to the packaging that helps cholesterol travel through the blood, and the molecular cholesterol that settles on blood vessel walls can even be cleared away. Part of the trouble begins when crystals of cholesterol form. These are the material of the plaques that form in lesions, causing irreversible damage that leads to atherosclerosis, and eventually to the heart disease and stroke that are the leading killers in the Western world. In research findings recently published in the Proceedings of the National Academy of Sciences (PNAS), a team at the Weizmann Institute of Science and their colleagues showed that these harmful cholesterol crystals form in two different ways, in immune cells that unwittingly aid in the progression of the disease.
When levels of LDL in the blood are too high, some of it is deposited on the blood vessel walls. There, the cholesterol undergoes a chemical change – oxidation – which turns these molecules toxic to the cells lining the blood vessel walls. The immune system, recognizing the danger, sends in its clean-up crew: the “Pacman”-like cells called macrophages that “eat” unwanted stuff in the body. But these cells can overeat: The stuffed macrophages become inflated “foam cells” that soon die. So at the site of the original lesion, there is a buildup of the remains of dead foam cells and escaped fatty molecules, exacerbating the inflammation and leading to the formation of the cholesterol crystals.
Prof. Lia Addadi of the Institute’s Structural Biology Department says: “The buildup of cholesterol crystals is a major step in a pathological chain of events that includes cell death, inflammation and the onset of arterial disease. We find that the crystals can form very early on in this chain.”
Dr. Neta Varsano in Addadi’s lab, who led the research, says: “Crystals are observed outside of the macrophages but also inside them, and it has been difficult to understand precisely when these begin to form.” She and members of the research team, in a long collaboration with the lab of Prof. Leslie Leiserowitz of the Institute’s Materials and Interfaces Department, set out to devise a method that would enable them to trace the process.

To observe the crystallization process in the lab, the team grew macrophage cells and added LDL cholesterol to them. These were then observed with a combination of methods that included high-resolution fluorescent microscopy and a type of cryogenic X-ray imaging that enabled them to get detailed, three-dimensional images of the crystals and follow their early growth.
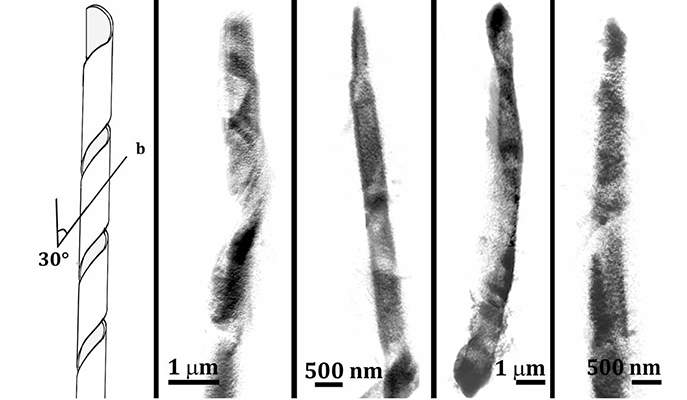
The earliest crystal structures to be detected were around 200 nanometers across, and these were thin, flat rhomboid-shape platelets that were found in the cells’ membranes. But then they discovered a second type of crystal – elongated, needle-shaped and tens of microns in size – inside the cells. These appeared, indeed, to act like needles that prick the cell membrane and rupture it. Addadi says that these crystals were a surprise, and they don’t show up in the standard test-tube studies of cholesterol crystals.
Together with Dr. Nadav Elad of the Institute’s Chemical Research Support, they characterized the crystalline structure of each formation. Each of these crystals had a different internal structure – the flat ones had a so-called triclinic crystal structure, while the elongated ones had a monoclinic structure. They noted that the elongated crystals were organized around a hollow or cylindrical tube, or they were, surprisingly, helical. Similar crystals had been seen in relation to gallstone formation.
These crystals don’t show up in the standard test-tube studies of cholesterol crystals
Varsano: “Even if their internal structures undergo changes, we think that the crystals preserve their original shape as they continue to grow in the arterial lesions. This means that we can look at the crystals in a sample and understand how and when they formed.” More work is needed to completely solve the riddle of how this helpful substance turns to killer crystals, but the research team is continuing to work on the experimental model – for instance, comparing samples from different humans to see if the crystals form in the same way in everyone or if there are variations.
Particpating in this research were Drs. Tali Dadosh and Iddo Pinkas of the Institute’s Chemical Research Support; Fabio Beghi of the University of Milan; Drs. Eva Pereiro and Ana Perez-Berna of the ALBA synchrotron in Spain; and Prof. Howard Kruth of the Experimental Atherosclerosis Section, National Heart, Lung, and Blood Institute, National Institutes of Health and members of his group.
Prof. Lia Addadi has had a long and fruitful career investigating the various ways that crystals work in living creatures. She was recently elected a member of the American Academy of Sciences, and this is her inaugural paper to the Proceedings of the Academy of Sciences (PNAS). “This work integrates all of my research methods and all of my thinking until now. It combined techniques from organic chemistry and crystallography with cutting-edge imaging technologies and, of course, close collaboration with my colleagues who are experts in the various aspects of this work.”
Prof. Lia Addadi is the President’s Advisor for Advancing Women in Science. Prof. Addadi is the incumbent of the Dorothy and Patrick Gorman Professorial Chair.