Misguided emergency measures can sometimes lead to disaster. In a study published recently in Cell Reports, Weizmann Institute scientists discovered that this may be happening in the rare genetic disorder called Batten disease. A subset of the patient’s brain neurons may be “thinking” they are subjected to starvation when in fact they are not, so they start taking unnecessary emergency measures that ultimately result in a fatal loss of brain cells. uided emergency measures can sometimes lead to disaster. In a study published recently in Cell Reports, Weizmann Institute scientists discovered that this may be happening in the rare genetic disorder called Batten disease. A subset of the patient’s brain neurons may be “thinking” they are subjected to starvation when in fact they are not, so they start taking unnecessary emergency measures that ultimately result in a fatal loss of brain cells.
This research began with a chance finding. A student in the laboratory of
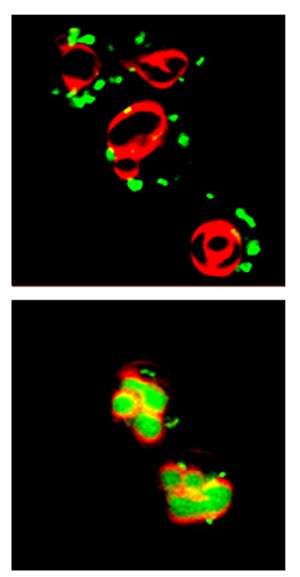
Prof. Jeffrey Gerst of the Molecular Genetics Department accidentally left some yeast cells overnight in a laboratory dish without their usual supply of nutrients. In the morning, the scientists found that the starved cells had undergone surprising changes. An organelle called the Golgi apparatus had altered its appearance and essential parts of this organelle had moved to the cell’s recycling machinery in order to be degraded. As a result, the Golgi, which normally dispatches proteins to their various locations within the cell, stopped working properly.
After investigating this phenomenon in depth, Gerst’s team – Niv Dobzinski and Rachel Kama – in collaboration with departmental colleagues Dr.Silvia Chuartzman and Prof.
Maya Schuldiner, discovered a previously unknown mechanism of cellular response to emergency. They found a direct connection to a signaling enzyme called TOR, a master protein switch that controls numerous cellular functions and serves as a monitor of the cell’s nutritional status. When the cell has adequate levels of nutrients, TOR sends the Golgi apparatus a “green light” signal to perform its usual role of “trafficking” proteins to different places within the cell. But when the levels of nutrients drop, TOR no longer sends out its signal. As a result, the cell enters an emergency shutdown mode, in which all non-vital processes – such as the functioning of the Golgi – are suspended, to free up the little available resources for synthesizing crucial proteins thus ensuring cellular survival. This cellular response to starvation goes into effect immediately, whereas other emergency measures may take several hours to be launched.
The scientists then realized that this speedy emergency response closely resembles the cellular phenomena they had observed in the past in a yeast model of Batten disease, a neurodegenerative genetic disorder that leads to gradual impairment of all functions and kills the patients by their twenties. Gerst’s lab, which focuses on investigating the movement of proteins within the cell, had unraveled the molecular mechanism behind this disease: In their yeast model, the scientists had seen that protein retention in the Golgi apparatus was disrupted. By extrapolation to human cells, they suggested that in Batten disease, certain proteins in brain neurons fail to be delivered to their correct destination, accumulating instead in the neuron’s recycling machinery and eventually helping to kill the cell.
The new study adds a new piece to the puzzle – by revealing that the disruption caused by the Batten disease gene is strikingly similar to the TOR-controlled emergency mode triggered by starvation.
This similarity suggests that due to a genetic mutation, affected neurons in Batten disease do not transmit the TOR signal and thus, mistakenly enter a starvation emergency mode. If this indeed proves to be the case, the search for a potential new cure for Batten disease may focus on restoring normal TOR signaling, to convince the patient’s neurons that they are not threatened with starvation after all.
Prof. Jeffrey Gerst’s research is supported by the Jeanne and Joseph Nissim Foundation for Life Sciences Research; the Miles and Kelly Nadal and Family Laboratory for Research in Molecular Genetics; the Yeda-Sela Center for Basic Research; the National Contest for Life (NCL)-Stiftung, Hamburg, Germany; the Israel Science Foundation; and the State of Lower Saxony, Hannover, Germany. Prof. Gerst is the incumbent of the Besen-Brender Professorial Chair of Microbiology and Parasitology.
Prof. Maya Schuldiner's research is supported by the Foundation Adelis; the Georges Lustgarten Cancer Research Fund; the Dora Yoachimowicz Endowed Fund for Research; the Berlin Family Foundation; Roberto and Renata Ruhman, Brazil; Karen Siem, UK; and the estate of Lola Asseoff.

 The new study adds a new piece to the puzzle – by revealing that the disruption caused by the Batten disease gene is strikingly similar to the TOR-controlled emergency mode triggered by starvation.
The new study adds a new piece to the puzzle – by revealing that the disruption caused by the Batten disease gene is strikingly similar to the TOR-controlled emergency mode triggered by starvation.