Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
Depression, schizophrenia, Alzheimer’s disease and other neurological and psychiatric disorders tend to strike one sex more than the other; and for reasons that remain unknown, they sometimes even produce different symptoms in women and men. A study comparing brain circuits in the two sexes of worms, conducted by researchers at the Weizmann Institute of Science, has identified a molecular mechanism that could help explain how the sex differences in human brain disorders come about.
The tiny, one-millimeter-long worm called Caenorhabditis elegans is an excellent model for studying brain circuitry: Its relatively simple nervous system contains a total of 302 neurons, and it’s the only organism whose entire set of neuronal connections in the brain has so far been mapped out for both sexes. At birth these connections are the same in the two sexes, but the circuitry starts to diverge when the worms mature sexually. Dr. Meital Oren-Suissa of Weizmann’s Neurobiology Department and her team thought that identifying the mechanisms of this divergence in the worm’s nerve circuitry could shed light on sex-related differences in the human brain, particularly the ones that may be linked to psychiatric disorders.
In experiments led by Dr. Yehuda Salzberg, the scientists focused on a particular connection between two neurons that allow the worm to sense its environment. This connection is known to be present in both sexes in juvenile worms, but as they mature sexually, it is retained in males but disappears in females. How does this difference in wiring emerge from a common template? (Side note: The female worms are also called hermaphrodites because they produce both eggs and sperm, but their nervous system is genetically female.)
By examining several potential mechanisms for the destruction of neuronal connections, Salzberg and colleagues established that in adult females, the connection in question is pruned, or removed, by means of a well-known molecular tag called ubiquitin. This tag marks proteins for breakdown and recycling in organisms all along the evolutionary tree, including humans; among its other functions, it takes part in shaping neuronal circuits during development.
Variants of the gene that encodes this receptor have been associated with major depression, schizophrenia, bipolar disorder and autism
Next, the researchers revealed the molecular details of the pruning. In experiments with mutant worms, they found that an enzyme called E3, which exists in humans as well, plays a central role in pruning the neuronal connections, as it leads to the attachment of the ubiquitin tag. The E3 enzyme comes in numerous versions, but the researchers managed to identify the precise one that carries out the ubiquitin attachment in the female worm’s nervous system. They then filled in other pieces of the ubiquitin puzzle. They found that when the E3 enzyme binds to a receptor known as DCC on the neuron’s outer membrane, the ubiquitin tag it adds marks the DCC receptor for destruction.
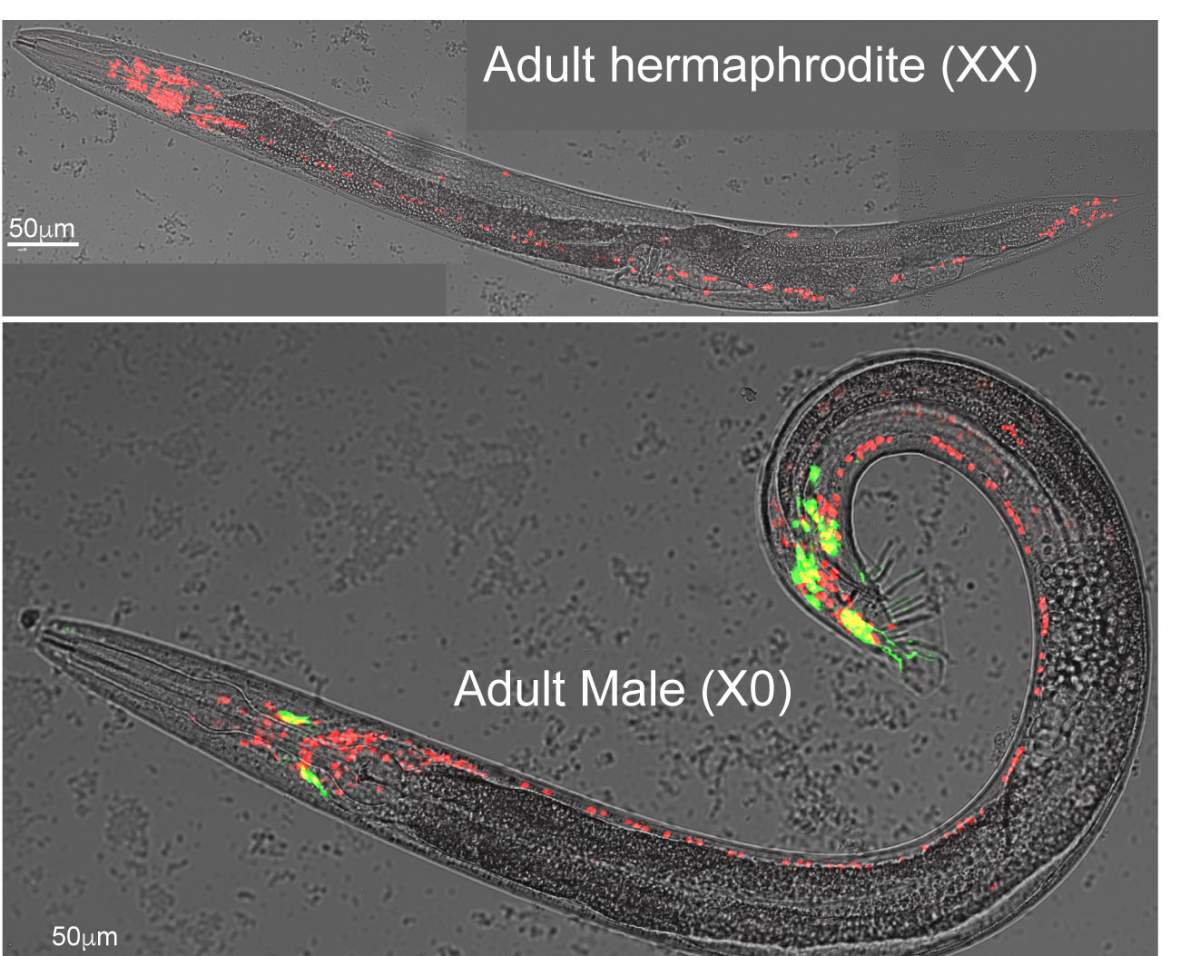
The researchers found that in males, neurons release a biochemical signal, netrin, that blocks the binding between the E3 and the DCC receptor, thereby protecting DCC from destruction. In female neurons, in which netrin is not expressed, the DCC receptor is bound by E3, tagged by ubiquitin and destroyed, ending in the pruning of the entire connection. When the scientists prevented the destruction of the DCC receptor in female worms, they saw the same brain circuitry as in males.
Could the DCC receptor hold an important clue to the origin of sex differences in brain disorders in humans? Certain variants of the gene that encodes this receptor have been associated with major depression, schizophrenia, bipolar disorder, autism and at least four other neuropsychiatric conditions – all of which differ in the prevalence and nature of symptoms in women and men. The new role for this receptor identified by the Weizmann scientists suggests a new direction for research that may help clarify the molecular mechanisms behind the sex differences in brain disorders. And once such mechanisms are understood, it may be possible to develop future therapies that would be specifically tailored to women or men.
Study participants included graduate students Vladyslava Pechuk, Asaf Gat, Hagar Setty and Sapir Sela from Oren-Suissa’s lab.
Dr. Meital Oren-Suissa, who grew up in Kiryat Ata and Haifa, made her first scientific discovery while working on her PhD in biology at the Technion – Israel Institute of Technology. She found that a protein responsible for fusing cells together is active in neurons, which don’t fuse; she later discovered that this protein helps to sculpt neuronal structure. These surprising findings sparked her interest in the nervous system. During her postdoctoral studies at Columbia University, she investigated sex differences in neuronal wiring that emerge during development.
In her lab at the Weizmann Institute of Science, established in 2018, Oren-Suissa uses the Caenorhabditis elegans worm as a model for studying sex differences in the brain. “Medical treatments for many psychiatric disorders are largely based on studies in male animals, and they don’t effectively account for the differences in these disorders between women and men,” she says. “Tracing sex differences in the brain down to the molecular level can greatly contribute to our understanding of these disorders.”
Oren-Suissa lives on the Weizmann campus with her husband, an aeronautics engineer, and their two daughters, aged 10 and four.
Dr. Meital Oren-Suissa's research is supported by the Abramson Family Center for Young Scientists; the Azrieli Foundation; the estate of Emile Mimran; and the European Research Council. Dr. Oren-Suissa is the incumbent of the Jenna and Julia Birnbach Family Career Development Chair.