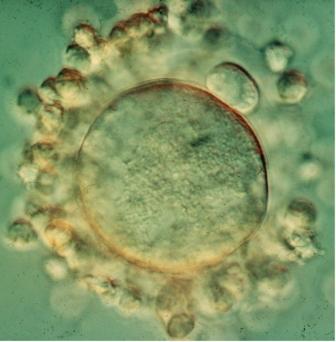
The series of studies that were ultimately to resolve this riddle began at the Weizmann Institute of Science in the early 1970s. Intrigued by the Pincus-Enzmann paradox,
Prof. Alex Tsafriri of the Biological Regulation Department, then a doctoral student, created the first system for culturing the large ovarian follicles of rats in a laboratory dish. This model proved useful for future studies of egg maturation in all mammals, including humans. It was thus possible to reveal the exact role of the ovulation hormone, LH. This was the first indication that a crucial molecular messenger called cyclic adenosine monophosphate (cAMP) plays a role in egg maturation.
This role was elucidated through the studies of several groups over many years, with crucial contributions by
Prof. Nava Dekel of the Weizmann Institute. In a nutshell, it turned out that LH triggers ovulation through a rise in follicle cAMP production. Yet in the egg, cAMP produces the opposite effect: A steady cAMP presence blocks egg maturation, which begins to proceed only when the level drops.
Tsafriri continued to study egg maturation upon returning to Israel and joining the Weizmann Institute faculty. In research spanning a decade, roughly between 1975 and 1985, Tsafriri and his colleagues from Baltimore, Profs. Channing and Seymour Pomerantz, came close to solving the Pincus-Enzmann paradox. As predicted earlier, OMI turned out to be a small peptide produced by follicle cells. Most importantly, by growing rat eggs and follicle cells together in test tubes, the scientists clarified how egg maturation is controlled: They suggested that follicle cells continually release OMI, blocking egg maturation, while LH overcomes this blockage. This explanation resolved the paradox in general terms: When the egg is placed in a laboratory dish, it matures without the hormone simply because OMI is no longer there to prevent it from doing so.
During a sabbatical at Stanford University, in collaboration with Prof. Marco Conti, Tsafriri identified the final link in this chain of events, the ultimate molecular switch that keeps egg maturation in check. This switch, an enzyme called PDE3A that degrades cAMP, is present in the egg itself. The scientists showed that as long as PDE3A remains in the OFF position, the egg does not mature, although other ovulation-related changes in the ovary can proceed undisturbed. These findings suggest that it might be possible to develop a method for blocking PDE3A as an optimal approach to contraception.
With time, it became clear that blocking egg maturation is a multi-step process. Why is such a complex molecular sequence needed to regulate egg maturation? “The sequence ensures that this process is carefully controlled, so that eggs don’t start maturing spontaneously by mistake,” says Tsafriri. “Egg maturation is so critical to life, it needs to be regulated with utmost precision, in concert with the changes that prepare the womb to be receptive to the embryo at the proper time.”
X-factor identified
As for OMI, the molecular “X factor,” it was finally identified recently by Prof. John Eppig and his group at the Jackson Laboratory in Bar Harbor, Maine, as reported in Science. Indeed, it’s a peptide, of the same size that Tsafriri and colleagues had predicted, called natriuretic peptide C (NPC); and indeed, it is continuously released by the follicle cells. A shutdown in NPC production unleashes the complex cascade of molecular events in the follicle cells that results in the activation of PDE3A (the ON position) in the egg, degradation of cAMP there, and eventually, egg maturation. “The discovery of this molecule and its action in the follicle has provided the ultimate proof for the OMI concept we formulated more than thirty years ago,” says Tsafriri.
The Bahat Prize
Prof. Alex Tsafriri was awarded the Bahat Prize given annually by the University of Haifa Press “for quality, original, non-fiction manuscripts in Hebrew that have not been published previously and which have a potentially large popular audience.” Tsafriri was awarded the prize for his work “Man as Animal?” (Ha-Adam K-Haya?), which provides a scientist’s perspective on the debates held in Israel and abroad over animal experimentation in biomedical research. Tsafriri discusses the moral and philosophical aspects of such experimentation, outlining how people-animal relations are treated by the different monotheistic religions, with an emphasis on Jewish theology. The manuscript will be co-published by the University of Haifa Press and Yedioth Books.