Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
A team of scientists from the Weizmann Institute of Science and the Hebrew University of Jerusalem has revealed the structure of a cellular editor that “cuts and pastes” the first draft of RNA straight after it is formed from its DNA template. Many diseases appear to be tied to mistakes in this process, and understanding the workings of the machinery involved may lead to the ability to correct or prevent them in the future.
Since the discovery, around 25 years ago, that the bits of DNA in the genes that code for protein formation are interspersed with “filler” segments that have no known function, scientists have worked to understand the process by which the right sequences are lifted out and strung together to make a coherent set of instructions. This act, referred to as “RNA splicing,” takes place in the “spliceosome” situated in the cell nucleus. A large complex of proteins and short strands of RNA, the spliceosome distinguishes the beginnings and ends of coded segments, precisely cutting and stitching them together. Alternative splicing, which underlies the huge diversity of proteins in the body by allowing segments of the genetic code to be strung together in different ways, takes place in the spliceosome as well.
The team consisted of husband-and-wife scientists Prof. Ruth Sperling of the Genetics department of the Hebrew University and Prof. Joseph Sperling of the Organic Chemistry Department of the Weizmann Institute, Ruth’s graduate student Maia Azubel, and Sharon Wolf of the Chemical Research Support Department at the Institute. They produced the most detailed 3-D representation of the spliceosome’s structure to date with their study, published today in Molecular Cell. Rather than follow previous attempts to unravel the workings of the splicing mechanism by studying spliceosomes created in test tubes, they managed to take spliceosomes directly from living cells and examine them under an electron microscope.
Their task was made difficult by the fact that spliceosomes in living cells are made up of four identical modules strung together like beads on a strand of RNA, each a miniature spliceosome capable of splicing on its own. The connections between the modules tend to be flexible, allowing the position of the units to vary in relation to each other. Thus pinning down a definitive shape and structure for the whole complex has been, until now, nearly impossible.
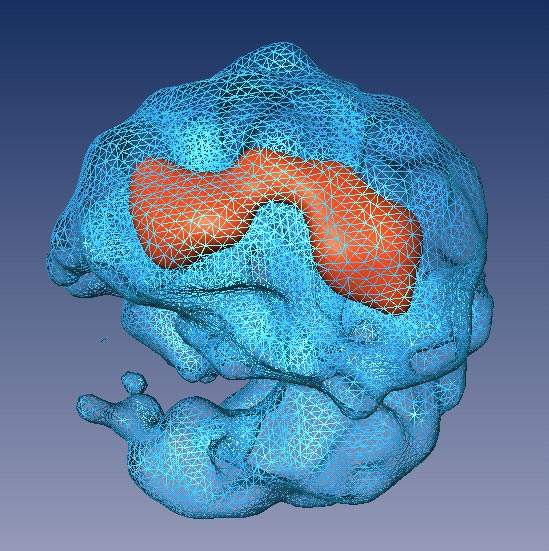
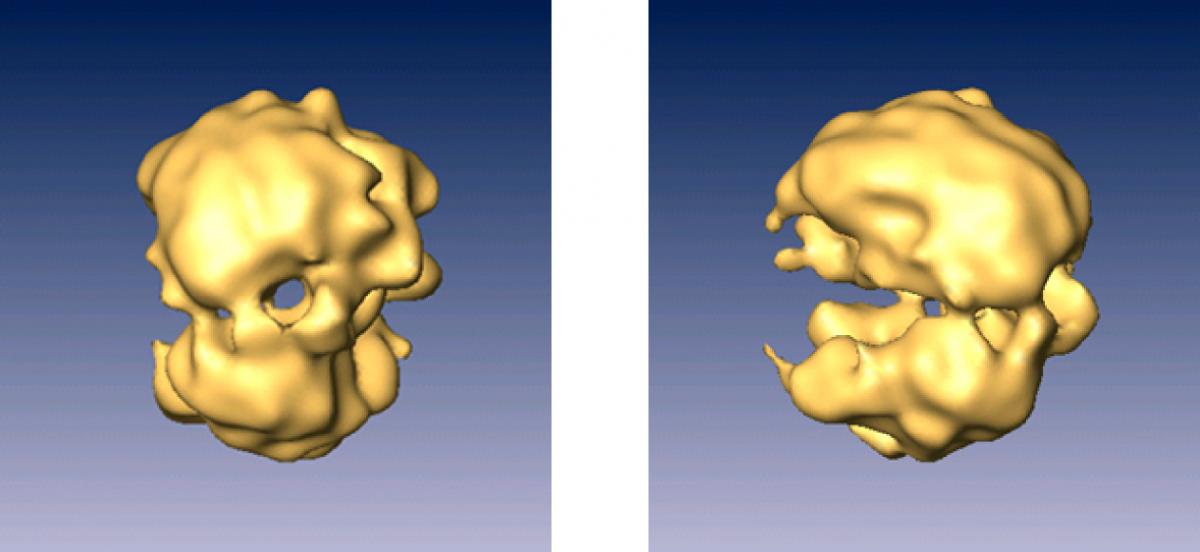
The team found a way to cut the RNA connections between the modules without harming the integral short strands of RNA that are essential to the splicing process, so they could study them individually. Split-second freezing at very low temperatures allowed the scientists to view the spliceosome units in as close to a natural state as possible. From thousands of images, each at a slightly different angle, a composite 3-D structure of the spliceosome was built up.
The revealed structure has two distinct, unequal halves surrounding a tunnel. The larger part appears to contain proteins and the short segments of RNA, while the smaller half is made up of proteins alone. On one side, the tunnel opens up into a cavity, which the researchers think functions as a holding space for the fragile RNA waiting to be processed in the tunnel itself.
What they didn’t see may be as important as what they saw. Whereas researchers examining splicing in test tubes saw evidence of a complicated sequence of events in which the spliceosome machinery assembles itself anew for each splicing job, the team’s investigations of spliceosomes from live cells found splicing to take place in pre-formed machines. This fits in with what is known about the way cells optimize their workload. “It’s much more efficient to have a machine on hand, ready to go, than to build a new one each time,” they noted.
Prof. Sperling's research is supported by the Joseph and Ceil Mazer Center for Structural Biology, the Helen and Milton A. Kimmelman Center for Biomolecular Structure and Assembly, the J. and R. Foundation, and Lois Zoller, Chicago, IL. He is the incumbent of the Hilda Pomeraniec Memorial Chair of Organic Chemistry.