The H.G. Wells classic War of the Worlds tells of a fictional invasion of Earth by evil Martians. Just when the Earthlings are about to give in to the aliens’ superior strength, the Martians pick up and leave, defeated by the tiny bacteria and viruses they encounter on Earth. When that book was written, humanity, itself, was engaged in a desperate struggle against the world of disease-causing microorganisms, which were still a leading cause of death. The tide began to turn only with the discovery of such antibiotics as penicillin. But bacteria have not admitted defeat; they have been developing resistance to our weapons at a pace that threatens to bring back the battle in full force. Since the first use of antibiotics, humans and bacteria have been engaged in an arms race in which the other side finds ways to overcome nearly any antibiotic we can develop.
Prof. Eitan Bibi and Ph.D. students Nir Fluman and Osnat Tirosh of the Biological Chemistry Department recently investigated this phenomenon. In two parallel studies on the MdfA transporter of the
E. coli bacterium, they revealed the mechanism for binding a proton and the conditions necessary for its release. The scientists then continued their study using genetic engineering techniques to create mutated proteins with the ability to bind and release more than one proton when ejecting an antibiotic molecule.
Under a microscope, the cell membrane appears as a thick, nearly impenetrable wall of lipids (fat-like substances) with embedded proteins (like MdfA itself) that span it from the inside to the outside.
The first study, which appeared in
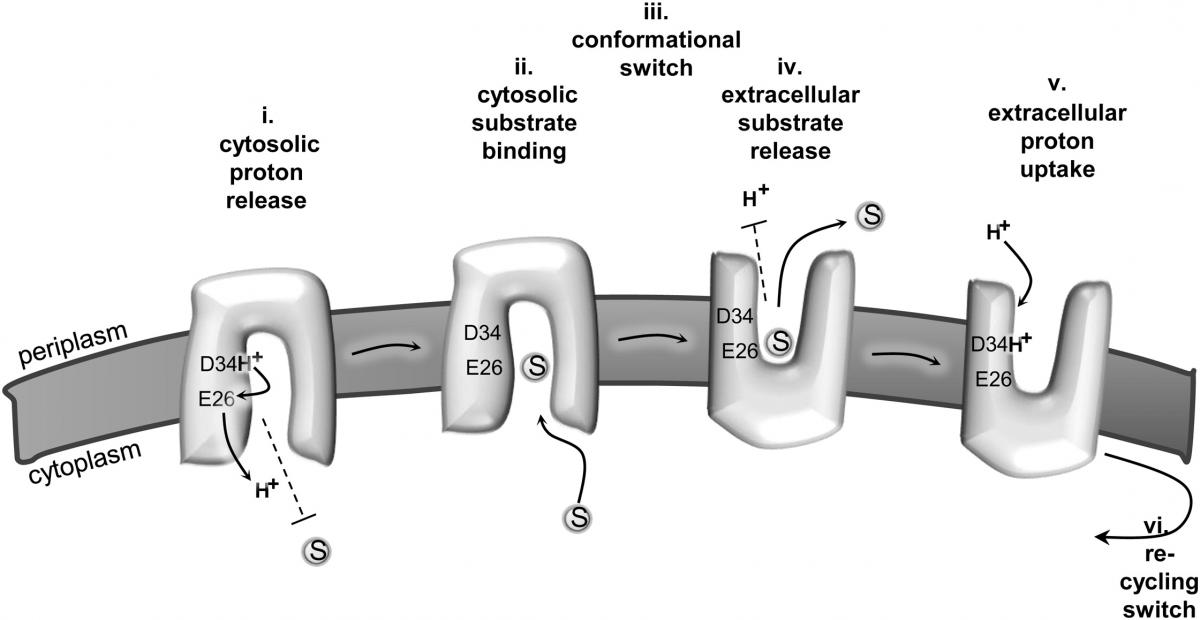
Molecular Cell, identified a site in the part of MdfA that is within the membrane – an acidic site (that is, it carries a negative charge) that binds the protons and releases them. This acidic site seems to act as a way station for protons passing between the membrane’s two sides. The research findings showed that this acidic site senses when an antibiotic is bound to the protein, releasing the proton into the cell when antibiotic binding takes place. In other words, getting rid of the antibiotic and taking in the proton are orchestrated actions. The mechanism works in the opposite direction as well, so that the binding of the proton causes the transporter protein to release its bound antibiotic to the outside of the cell. The scientists found that this phenomenon, known as competitive binding, is crucial for MdfA activity. But they were surprised to discover that the antibiotic and the proton bind at different locations in MdfA, with the information passed between the two binding sites. Processing this information causes the protein to undergo a structural change, and this, in turn, leads to the antibiotic’s ejection.
This resistance mechanism works when the antibiotic molecules are exchanged for one proton, but does not help against certain substances that require the import of two protons. In
the second study, Bibi and his students genetically engineered bacteria to be resistant to these substances, as well. That is, the transporters could transfer two protons instead of one. The researchers introduced genetic mutations into MdfA and found several that grant resistance. Interestingly, each of the mutated transporters had an additional acidic site created near the original proton-binding site.
This appearance of a second proton-binding site in the mutant transporters suggests that the proton-transfer mechanism might be modular: By adding a second acidic site in the appropriate location, the protein gained the ability to transfer two protons at once. These findings, which appeared in the Proceedings of the National Academy of Sciences (PNAS), shed new light on the potential ease with which bacteria can develop antibiotic resistance.
This research is especially important right now, as antibiotic resistance is growing and new discoveries in the field are slowing down. A deeper understanding of the proteins that remove antibiotics from the cell will aid humanity in its eternal arms race against disease-causing bacteria.
Prof. Eitan Bibi's research is supported by the Dr. Josef Cohn Minerva Center for Biomembrane Research, which he heads; the Edmond J. Safra Philanthropic Foundation; the Willner Family Leadership Institute for the Weizmann Institute of Science; the Jeanne and Joseph Nissim Foundation for Life Sciences Research; Rudolfine Steindling; the estate of Harold Z. Novak; the estate of Irwin Mandel; and the estate of Raymond Lapon. Prof. Bibi is the incumbent of the Ruth and Jerome A. Siegel and Freda and Edward M. Siegel Professorial Chair.