Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
Immune cells zip through our body’s blood vessels at dizzying speed, braking at critical checkpoints with the precision of a plane landing on the deck of an aircraft carrier. This remarkable ability enables them to exit the vessels at just the right spots to cross into any site of injury or into lymph nodes, which they continuously patrol for signs of infection. But how do these immune cells detect such exit sites with extraordinary precision? Weizmann Institute of Science researchers set out to find the answer; they learned that key immune cells called T lymphocytes keep the braking machinery right at their “fingertips.”
We tend to picture cells as smooth sacs, yet the surfaces of many cell types – and certainly those of all immune cells, including the T lymphocytes that patrol the lymph nodes – are anything but even. Their outer membranes have elastic finger-like projections called microvilli that help the cells sense and interact with their environment. When a T lymphocyte needs to cross the wall of a blood vessel in order to get inside a lymph node, receptors on its “fingers” bind to specialized proteins on the vessel wall. Once fastened in this manner, the cell starts rolling along the vessel walls. As it does so, its receptors called CCR7 recognize small lymph node proteins called chemokines, which coat the inner surface of blood vessel walls. The chemokines let the T cell know: You’ve arrived at the right spot in a lymph node – time to get off.

Only cells that have CCR7 receptors can read this exquisitely targeted message. The CCR7 transmits the signal further, activating an adhesion protein called LFA-1 integrin, which in turn binds to sticky molecules on the blood vessel wall. The T cell can then come to a sudden halt, exit the vessel and enter the lymph node, where it spends anywhere from a few minutes to several hours on the lookout for foreign particles, before returning to the blood circulation if it encounters nothing troublesome. Thousands of such events take place within hundreds of lymph nodes around our body every minute, ensuring effective immune surveillance.
In earlier research, Prof. Ronen Alon and his team at Weizmann’s Immunology Department had found that the braking signal in the T cell fingers is generated within less than half a second – a rapid time frame that was puzzling on several fronts. First of all, each second a T cell moves through the blood vessel, it covers a distance of about a tenth of a millimeter – that is, about fifteen times its diameter – a pace that in the macro world would be roughly equivalent to the speed of a landing airplane. But when an airplane lands on a narrow flight deck, steel cables meant to arrest its movement are already in place, whereas each time a T cell “lands” in a lymph node, it assembles all the braking machinery anew on the tips of its microvilli. Not only that, a crucial component of this machinery, the LFA-1 integrin, was in the past found largely on the cell body, far away from the tips of microvilli.

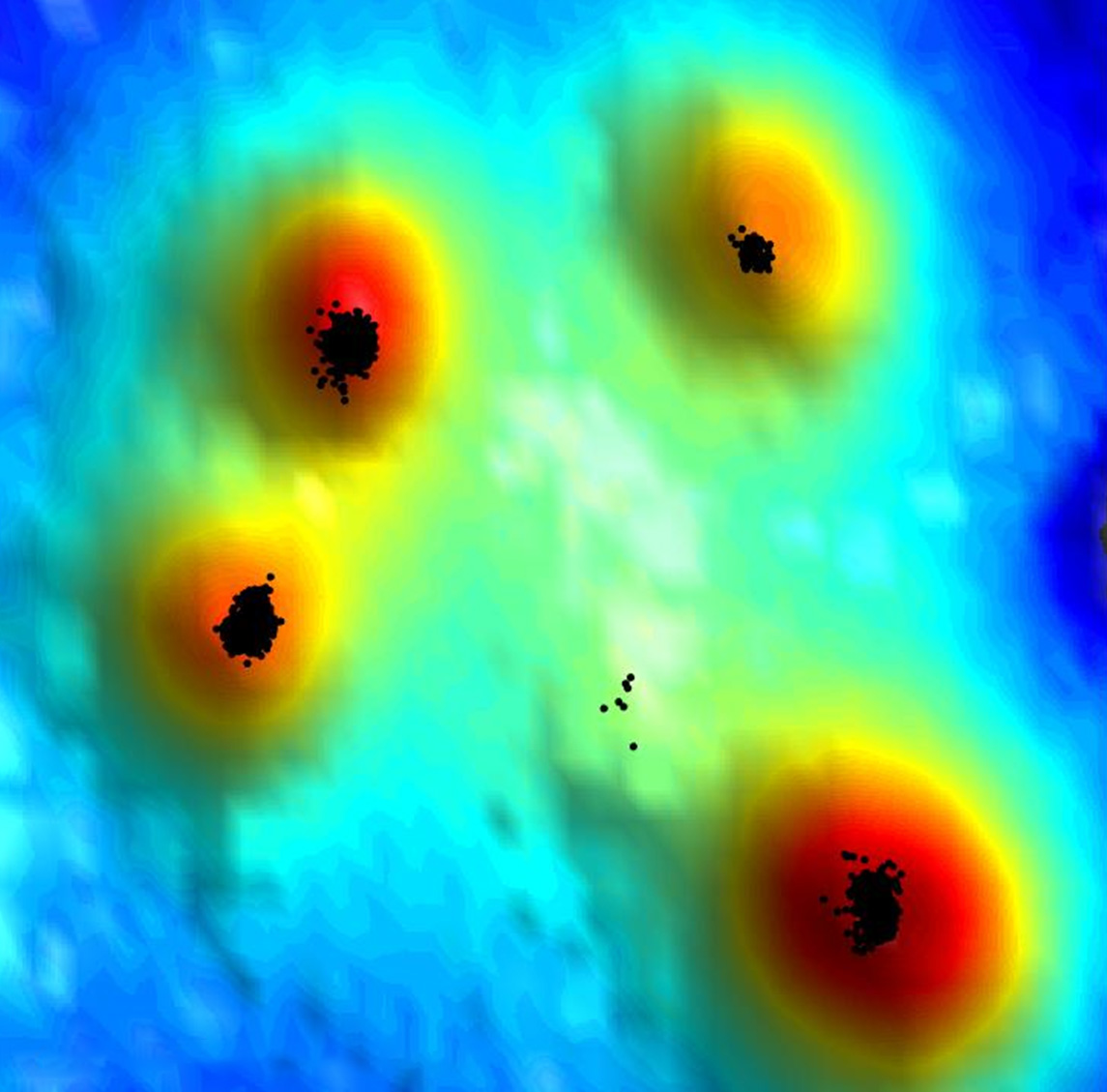
To learn how T cells manage to put together their braking machinery so quickly, Alon teamed up with Prof. Gilad Haran of the Chemical and Biological Physics Department, whose lab has developed a new super-resolution microscopy method for exploring cell surfaces at the level of single molecules. It combines two techniques: One determines the location of various molecules stained by fluorescent markers; the other gages the intensity of the fluorescence signal to create a topographic map of the cell surface, including the microvilli. “Our method maps out the ‘hills’ and ‘valleys’ of the cell surface, similarly to the way a topographic map presents the shape of the earth’s terrain,” Haran says.
""All the machinery that T cells need for their rapid braking is, in fact, pre-assembled on the tips of their microvilli, which explains how they manage to transmit the braking signal within less than half a second"
Postdoctoral fellow Dr. Shirsendu Ghosh from Haran’s lab and Dr. Sara Feigelson from Alon’s lab led the new study, in which they revealed how the T cells pull off their braking trick. The researchers sought to determine where the various components of the T cell braking machinery are located and found that the CCR7 receptors are clustered in the most accessible spot, right at the tips of the microvilli. No less important, it turned out that some of the T cell’s LFA-1 molecules are conveniently available nearby: Although they are indeed mostly found on the cell body, the researchers identified a subset making up about 5 percent of the LFA-1 that is dispersed along the surface of the microvilli, including their tips. Finally, and equally important, all the assistant signaling molecules needed for assembling the braking machinery were also found to be clustered very close to the CCR7 at the tips of the T cell microvilli.
“All the machinery that T cells need for their rapid braking is, in fact, pre-assembled on the tips of their microvilli, which explains how they manage to transmit the braking signal within less than half a second,” Alon says.
In addition to shedding new light on the way T cells perform immune surveillance of the body, these findings open up a new direction of research into mechanisms guiding the movement of other cell types, including tumor cells that, while flowing through blood vessels, come to a halt at sites of metastases. Furthermore, the findings may one day help develop ways of manipulating the movement of immune cells on demand. For example, it may be possible to enhance the effectiveness of vaccines by increasing the number of CCR7 receptors on the microvilli in order to enhance the immune cells’ ability to arrive at lymph nodes, or, conversely, to restrict such arrival in order to reduce the risk of autoimmune diseases.
Study participants included Dr. Alessio Montresor and Prof. Carlo Laudanna, from the University of Verona, Italy; Dr. Eyal Shimoni, from Weizmann’s Chemical Research Support Department; Dr. Francesco Roncato from Weizmann’s Immunology Department; and Prof. Daniel F. Legler from the Biotechnology Institute Thurgau, Kreuzlingen, Switzerland, of the University of Konstanz.
The average diameter of a T cell circulating in blood is 6-7 microns; its microvilli measure from 100 nanometers to 0.5 micron in length; every T cell surveys about 10 different lymph nodes per day.
Prof. Ronen Alon is the incumbent of the Linda Jacobs Professorial Chair in Immune and Stem Cell Research.
Prof. Alon's research is supported by the Moross Integrated Cancer Center; and the Yeda-Sela Center for Basic Research.
Prof. Gilad Haran is the incumbent of the Hilda Pomeraniec Memorial Professorial Chair.
Prof. Haran's research is supported by the Henry Chanoch Krenter Institute for Biomedical Imaging and Genomics; the Nancy and Stephen Grand Center for Sensors and Security; the André Deloro Institute for Advanced Research in Space and Optics; and the Harold Perlman Family.