“It’s all in your head” is more than a catchy phrase; it’s a fairly accurate description of how the human body functions. From blood pressure to appetite to food metabolism, much of the body’s chemistry is controlled by the brain. In
a study published recently in
Developmental Cell, a team of scientists led by
Dr. Gil Levkowitz of the Weizmann Institute has revealed the exact structure of one crucial brain area in which biochemical commands are passed from the brain cells to the bloodstream, and from there to the body.
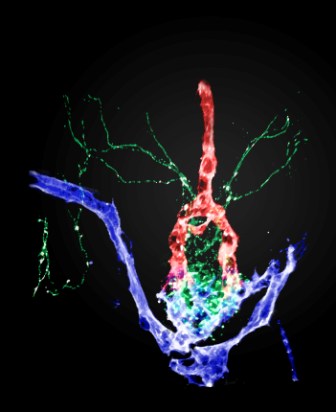
The brain area in question, the neurohypophysis, is located at the base of the brain, where it is an interface between nerve fibers and blood vessels. Here, some of the major brain-body interactions take place: the nerve cells release into the blood vessels the hormones that regulate a series of vital body processes, including the balance of fluids and uterine contractions in childbirth. The neurohypophysis has been studied for more than a century; but now, in the Weizmann Institute-led study, the scientists have developed new genetic tools that allowed them to re-examine the exact three-dimensional arrangement of this brain structure, along with clarifying the cellular and molecular processes leading to its formation.
Since the human neurohypophysis, composed of tens of thousands cells, is exceedingly complex, the scientists performed the research on live embryos of zebrafish, which offer a unique model for studying the vertebrate brain. As these embryos are entirely transparent and lend themselves to genetic manipulation with relative ease, the scientists could observe the actual formation of the fish neurohypophysis under a microscope. The findings apply to humans because this part of the brain is built similarly in all vertebrates. The research was conducted in Dr. Levkowitz’s lab in the Molecular Cell Biology Department by Ph.D. student Amos Gutnick together with Dr. Janna Blechman. The Weizmann scientists worked in collaboration with Dr. Jan Kaslin of Monash University, Australia; Drs. Lukas Herwig, Heinz-Georg Belting and Markus Affolter of the University of Basel, Switzerland; and Dr. Joshua L. Bonkowsky of the University of Utah, United States.
The study revealed a surprising new function for the hormonal messenger oxytocin, dubbed the “hormone of love” because in addition to controlling appetite (see box) and female reproductive behaviors such as breast-feeding, it is also involved in mother-child and mate bonding. The scientists have shown that oxytocin, one of the two major hormones secreted in the adult neurohypophysis, is involved in the development of this brain area already at the embryonic stage. In the embryo, oxytocin released by nerve cells governs the formation of new blood vessels. “In this manner,” says Levkowitz, “the messenger helps to build the road for transmitting its own future messages.” Developmental Cell highlighted the study’s findings in a preview headlined, “The Hormone of Love Attracts a Partner for Life.”
These findings provide an important advance in basic research because they shed light on fundamental brain processes, but in the future they might be relevant to the treatment of disease. Since the neurohypophysis is one of just a few portions of the brain that regenerate after injury, understanding how it is formed may one day help achieve such regeneration in other types of brain injury.
Appetite Tidbits
Among its numerous functions, the hormone oxytocin suppresses appetite. It would seem that oxytocin could make a fantastic diet pill: Encourage the body to make more oxytocin, and your hunger will go away. Alas, if only things were so simple!
Weizmann Institute researchers have found that a protein called PGC-1alpha, which stimulates energy expenditure due to fasting, exposure to cold or exercise, also stimulates the production of oxytocin. In other words, the same molecule triggers two biochemical processes that have exactly opposite effects: one increases appetite by enhancing energy expenditure, the other suppresses the appetite.
This study, reported recently in the
Journal of Neuroscience, was performed in Dr. Levkowitz’s lab by Dr. Janna Blechman together with Dr. Liat Amir-Zilberstein and Amos Gutnick, as well as Dr. Shifra Ben-Dor of Biological Services. Using the zebrafish as a model, the scientists developed a unique genetic approach that allowed them to clarify how PGC-1alpha affects the release of oxytocin by the brain’s nerve cells.
If these findings are found to be relevant to humans, they suggest that in order to manipulate appetite with the help of oxytocin, researchers would have to take into account the complexity of the regulatory mechanisms involved. In particular, they would need to separate the two opposing functions of PGC-1alpha, enhancing only the one that results in appetite suppression.
Dr. Gil Levkowitz's research is supported by the Dekker Foundation; the estate of Lore Lennon; the Kirk Center for Childhood Cancer and Immunological Disorders; and the Irwin Green Alzheimer's Research Fund. Dr. Levkowitz is the incumbent of the Tauro Career Development Chair in Biomedical Research.