As we get older, our memory tends to get creaky. According to new research, this “rusty brain” could be tied to an aging immune system. Two recent studies in the lab of
Prof. Michal Schwartz of the Institute’s Neurobiology Department reveal how certain immune cells – located in an unusual site, right on the edges of the brain – benefit the central nervous system (CNS), among other things, in healing injury. But they also suggest that a shift in the border’s gateway over time can contribute to declining mental function and neurodegenerative disease. These studies contain clues as to how we might, in the future, be able to slow or prevent cognitive decline and fight neurodegenerative conditions.
Following their discovery over a decade ago that circulating immune cells play a role in brain and CNS function, Schwartz and her group have been investigating the underlying mechanism. This research has often challenged the conventional wisdom: It was believed that immune cells were not allowed to infiltrate the brain, and if they did, their entry was considered to take place through fractures in the brain’s barrier system.
The results of
the first study, which appeared recently in
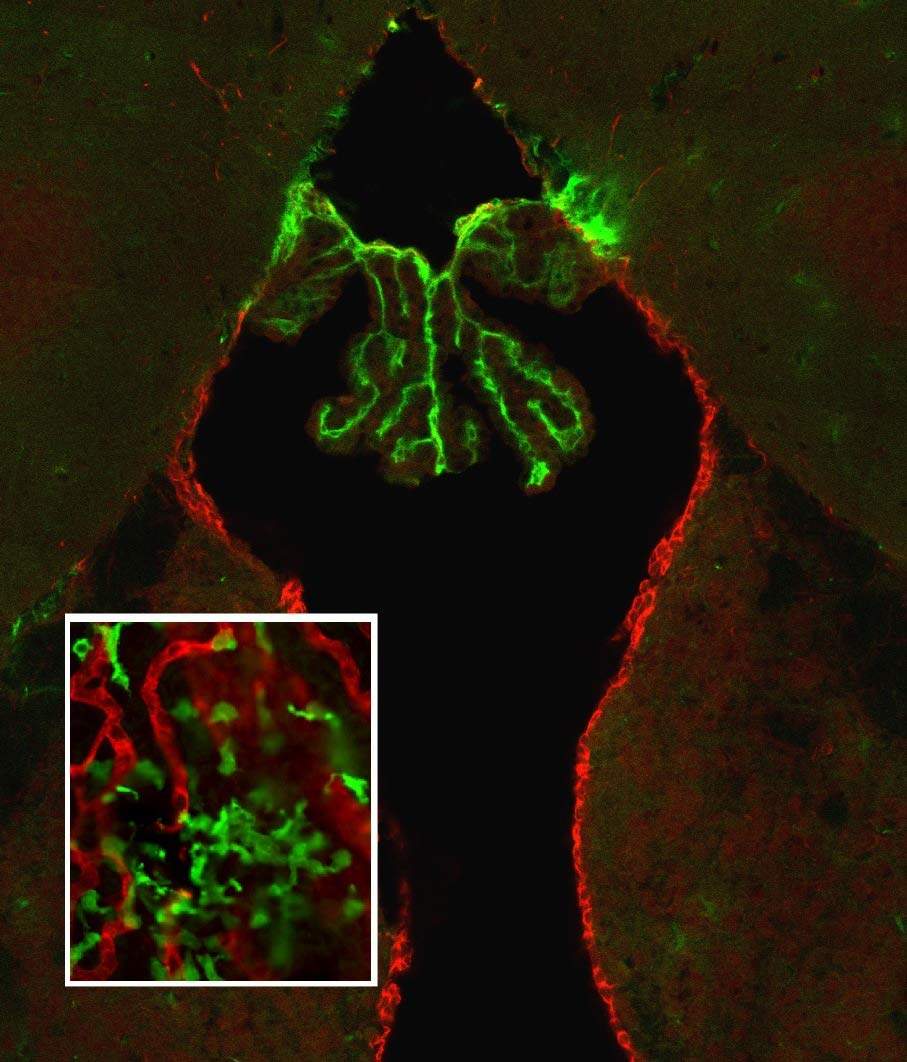
Immunity, showed that when healing immune cells are summoned to the brain following trauma, they do not need to breach barriers. Schwartz’s team discovered that the healing cells enter through structures within the brain called choroid plexi, which form the so-called cerebrospinal-fluid barrier. These structures – one choroid plexus in each quarter of the brain – have finger-like projections that exchange cerebrospinal fluid with the surrounding brain on the one hand and blood plasma and waste products with the blood vessels on the other.
To their surprise, the researchers found that cells needed for the repair of a distant spinal cord injury travel though this remote gateway, rather than drifting across breaches in the blood-brain barrier. The healing immune cells were found to cross the cerebrospinal-fluid barrier between the blood circulation and brain tissue and, from there, make their way to the injury site in the CNS. In other words, the cerebrospinal-fluid barrier, which was assumed to be as impenetrable as the blood-brain barrier, suddenly appeared to be quite porous. “It is more of a filter or a gate,” says Schwartz. “It sends immune cells through that gate when they are needed, though it first performs a security check and ‘educates’ them, so as to ensure that the right cells – with the right training – enter the system at the right time.”
In
the second study, which appeared in the
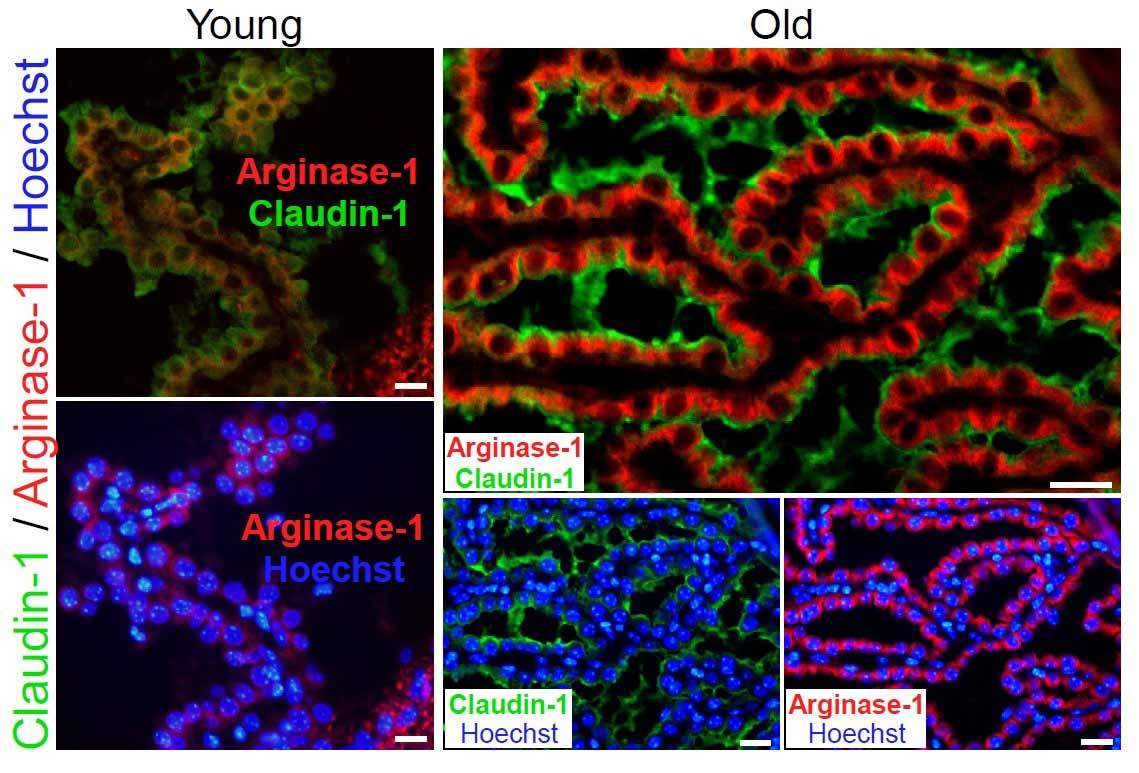
Proceedings of the National Academy of Sciences (PNAS), the team wanted to find the immune cell “gatekeepers” controlling this newly discovered compartment of the immune system. The research team identified the resident immune cells in the choroid plexus as a type of T cell that is specially adapted to the CNS, and found that these cells are kept permanently within the choroid plexus structures. A careful examination of cell fate from youth to old age revealed that changes in certain substances produced by these cells affected the gateway’s properties. One of these substances, interleukin-4 (IL-4), was known to control inflammation and help keep the brain healthy.
Paradoxically, however, it was also known to be involved elsewhere in the body in the production of a biochemical known as CCL11, which, among other things, has been implicated in cognitive decline. Further experiments in mice and cell cultures revealed that IL-4 generally exists in this compartment in equilibrium with another substance, interferon gamma. In the aging animal brain, changes in the general T cell populations shift the ratio in favor of IL-4, and this imbalance leads to inflammation similar to that seen in an asthmatic lung. The result is the production of CCL11 by the choroid plexus; from there, it enters the cerebrospinal fluid, where it accumulates and impairs cognitive ability in aged mice, and also in humans.
The research team managed to partially reverse the cognitive decline of the aged mice by applying a somewhat drastic treatment: They “reset” the mice’s immune system by irradiating their bone marrow from the neck down and transplanting new bone marrow. But Schwartz believes the findings contain hints that less radical means of restoring the immune balance in the brain may be feasible in the future. For one thing, the inflammation-causing T cells found in the aging choroid plexus are also present in elevated levels in the blood of the elderly, which suggests that a general strategy for targeting these cells might be helpful. For another, the intriguing similarities between the inflammatory process in the brain and asthma – which, says Schwartz, “is another disease of a filter, in this case, the airways”– suggesting further possibilities for treatment. And, as these findings make clear, the “barriers” separating the brain from the rest of the immune system are by no means impermeable if the right approach to the gate can be found.
In other words, rather than an inevitable side effect of aging, cognitive decline and age-related neurodegenerative diseases might one day be considered treatable conditions, like any other disease of the immune system.
Prof. Michal Schwartz’s research is supported by the Adelis Foundation; and the European Research Council. Prof. Schwartz is the incumbent of the Maurice and Ilse Katz Professorial Chair of Neuroimmunology.