Like a sprouting thicket, the cells of the developing nervous system extend long, thin branches called axons into all parts of the growing body. This thicket, at second glance, turns out to be a well-tended garden: As development progresses, quite a few extraneous nerve cells are weeded out and many of the axons are pruned back. This gardening activity remodels the nervous system even as it continues to spread and grow. It is the key to a well-functioning nervous system, and there is some evidence that certain neurological disorders, for instance autism, may be tied to imperfect remodeling.
Dr. Avraham Yaron and research student Maya Maor-Nof of the Weizmann Institute’s Biological Chemistry Department
recently revealed a central player in the process of axon pruning. This is crucial, says Yaron: “Before we can understand the mechanisms involved in health or disease, we must be able to identify the various players involved. Until now, we have had very little idea of who or what these players are.”
Pruning of axons takes place from the inside out: Long structural elements, called microtubules, that run down their length are first disassembled. Microtubules are both backbone and transport system: various payloads, including signaling molecules and other materials, are conveyed up and down the rail-like microtubule system. The researchers wanted to know what mechanism controls the breakdown of microtubule structures during axon pruning. Working with sensory nerve cells grown in culture, they used a process of elimination to identify the one – called KIF2A – that carries out the job. Interestingly enough, KIF2A is a member of the family of motor proteins that travel up and down the microtubules and transport various cargos.
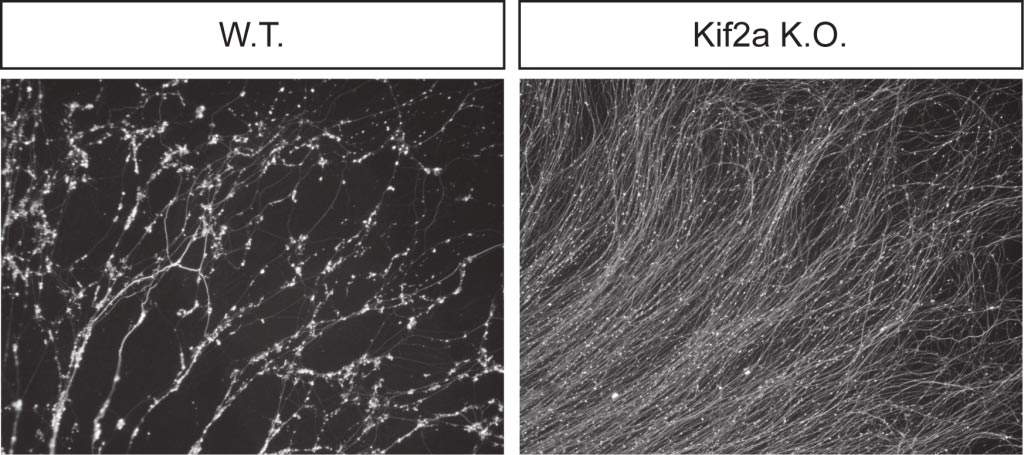
To understand the role KIF2A plays in nervous system development and axonal pruning, the Weizmann researchers obtained knockout mice from scientists from the University of Tokyo, Japan, that were missing KIF2A. In the KIF2A-deficient mice, the axons grew normally but the pruning process was impeded as the microtubules and the axons remained intact.
This confirmed that KIF2A is the crucial factor – one which must be present for pruning to occur. The KIF2A knockout mice were distinguished by the fact that their skin – a main destination for developing sensory nerve axons – was inundated with numerous nerve endings.
How does the process work? The research findings suggest that KIF2A is controlled by molecules called neurotrophins that play the role of head gardener. Neurotrophins are found in the nerve’s target tissues (the skin, for example, in the case of the sensory nerve cells in the experiments), where they guide the growth of axons in the right direction and help them to survive. But neurotrophin numbers tend to be limited, and the axons end up competing for their signals. When an axon does not obtain a neurotrophin signal, its KIF2A, which is normally present in an inactive form, will become activated and the pruning procedure will be initiated.
This creates a process, according to Yaron, that at first appears to be somewhat random but is in reality quite orderly. Not only are the numbers of axons carefully regulated – leading to optimum configurations of nerve cells – but the pruning also proceeds step by step in a preset, genetically determined order. If the first step – microtubule disassembly – does not take place, the rest of the pruning process cannot follow.
Previous research in Yaron’s group, conducted together with
Dr. Eli Arama of the Molecular Genetics Department, investigated another type of machinery that regulates nervous system remodeling – the apoptotic (cell suicide) system . Taken together, this research is beginning to fill in the details of how the nervous system’s “garden” grows: exuberant growth and development alongside judicious thinning, weeding and pruning. Once researchers understand where the balance between them lies and how that balance is achieved, they can begin to understand diseases that arise out of an imbalance, says Yaron. There are some indications, for instance, that autistic brains may suffer from too little pruning – leading to an excess of nerve connections. Yaron thinks that a faulty KIF2A mechanism may play a role in the disorder.
Prof. Avraham Yaron's research is supported by the Nella and Leon Benoziyo Center for Neurosciences; the Wolfson Family Charitable Trust; the Koshland Family; the Rowland and Sylvia Schaefer Family Foundation; the Irving Harris Foundation; the estate of Lola Asseof; the estate of Nathan Baltor; and the Joseph D. Shane Fund for Neurosciences.