Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:

Marine microorganisms called phytoplankton undergo some of the most extreme population swings on the planet – forming huge blooms that cover kilometers, then crashing to barely detectable levels within weeks. In the process, they provide some 50% of the planet’s oxygen; they make up a substantial part of the base of the marine food chain; and the delicate mineral shells they create end up everywhere from cloud droplets to the ocean floor. The Weizmann Institute of Science’s Prof. Assaf Vardi says: “Blooms of these photosynthetic single cells have a major impact on the carbon cycle in the ocean.” The blooms’ lifespan is limited by several factors conspiring against the algae – which may run out of nutrients, die from viral or bacterial infection or be eaten.
“The pattern – profligate cell division and rapid cell death – is the result of innumerable individual decisions within trillions of single cells,” adds Vardi, who is in the Institute’s Plant and Environmental Sciences Department. What separates the ones that survive from the others? How do cells survive in the face of multiple stressful conditions and go on to generate new blooms? To explore these issues, he and his team first developed new experimental techniques, designing microfluidic equipment that enabled them to continuously observe living cells under a microscope for a period of 24 hours, and to expose cells to a closely controlled environment in which various stresses could be applied. “Most research on microorganisms like phytoplankton is based on averaging out their behavior and response levels for the group as a whole. But such studies miss a lot of interesting, subtle strategies exhibited by individuals,” says Vardi.

Led by PhD student Avia Mizrachi, the group worked with a diatom, P. tricornutum, for which there is a known genetic toolkit with which to investigate their unique acclimation to stress. The group also focused on molecules known as reactive oxygen species (ROS) that play a role in the perception of environmental stress and which may activate one of the programs carried in the cell for either cell death or acclimation. High ROS levels (as opposed to the low levels needed for normal metabolism) damage cells and get them to commit suicide. But do all diatom cells react in the same way to its signal?
Right away, the researchers noted that when they exposed diatoms to moderate levels of ROS, the diatom cells formed two distinct groups: Many were quite sensitive to the ROS message and initiated their emergency cell death programs, whereas others appeared to be resilient – able to respond to the signal by acclimating. This split showed up even when the experiments were conducted on genetically identical, cloned diatom cells. In other words, even with genetically identical diatom cells that start out with the same chances of surviving, only a few will manage to make it through the taxing ordeal.
For more clues, the researchers zeroed in on the sites of ROS production in diatoms: the photosynthesizing organelles called chloroplasts, which harvest the sun’s energy and produce ROS as a byproduct. If their hunch was correct, the chloroplasts serve not just as machinery for converting carbon dioxide to sugar, but as participants in the decision-making mechanisms in the cell. Vardi and the research group inserted a so-called reporter – a sort of label that glows in fluorescent colors in response to ROS levels – into chloroplasts and other cellular organelles, and they followed the reporter as the diatoms were exposed to stress conditions that are prevalent at the ocean’s surface and are known to instigate increased ROS production.
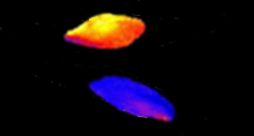
In the more intense light – like that on a sunny day at the ocean’s surface – a fair number of diatom cells accumulated ROS in their chloroplasts and died while others were, indeed, able to survive. Mizrachi found, however, that this split into two groups – sensitive and resilient – occurred only when they were in the light. After being kept in the dark, all the cells became sensitive to the ROS signals; and these provoked death in the entire population. “Diatoms need sunlight to survive, but too much of it may be deleterious for them. At the other end of the spectrum, without light they become very sensitive to other stressful conditions. It is possible that those floating just a bit deeper – trading slower growth in lower light conditions for slower ROS production in the chloroplasts – might be those that have a better chance of survival,” says Mizrachi.
The cells’ reactions were visible as soon as 15 minutes after the ROS molecules were produced, though the cells did not die for another 24 hours. Mizrachi found she could reliably predict – just by the initial reactions of the chloroplasts to the ROS signals – which cells would be dead a day later. That is, once these organelles reacted in a certain way to the ROS signal, the suicide plan was initiated and locked in place. Using this insight, the group is now working towards uncovering the genes and other signaling molecules that work to either obey or ignore the signals and ensure cell death or survival. These, says Vardi, are what turn the general ROS signal into one that is specific for the perils diatoms face and the responses each evokes. Mizrachi also developed a method for separating the two groups, a feat that is already enabling the research team to further probe the dynamics of death and survival. “We are continuing to explore exactly which factors determine survival or cell death, as well as which genetic programs mediate these decisions in each cell,” says Mizrachi. “Light exposure seems to be one such factor; others might include metabolism, the cell’s history, external signals or other aspects of their environment.”
ROS signaling told the researchers 24 hours ahead which diatoms would live and which would die. Microfluidics in vivo imaging of chl-roGFP oxidation over time following H2O2treatment
“Different interpretations of various messages – ROS is just one of many – probably evolved as a sort of ‘bet-hedging’,” she adds. “A way to ensure that no matter what, at least a small percentage of cells will survive to keep the species going.”
“These microorganisms have evolved a unique cycle in which ‘survival of the few’ enables the species, as a whole, to continue,” says Vardi. “In my lab, our challenge is to understand how the decisions that occur on the scale of single cells continually affect processes that take place on the global level. It is precisely the ability to look into those cells at this new resolution – really at the scale of what happens inside individual organelles within those cells – that is enabling us to do just that.”
Prof. Assaf Vardi's research is supported by the Willner Family Leadership Institute for the Weizmann Institute of Science; the David and Fela Shapell Family Foundation INCPM Fund for Preclinical Studies; the De Botton Center for Marine Science; the Edmond de Rothschild Foundations; the Bernard and Norton Wolf Family Foundation; Claire and Marc Perlman; Scott Eric Jordan; the estate of Emile Mimran; and the European Research Council.