Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
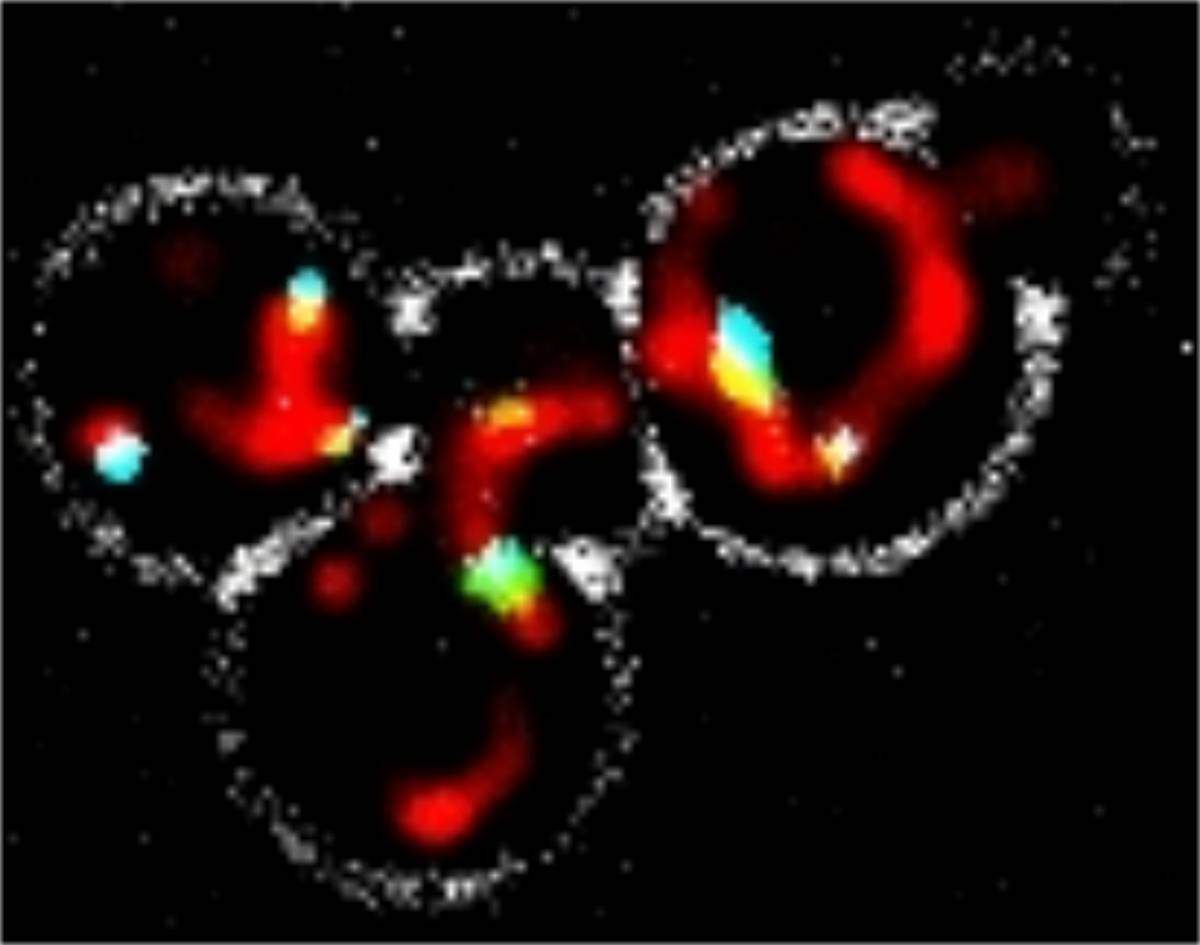
Reaching out and touching is so natural, even the organelles inside our cells do it. New research at the Weizmann Institute of Science used a unique method to uncover a number of new such areas of contact, or contact sites, between organelles as well as several tiny “tethers” that extend from one organelle’s membrane to another, enabling physical contact. Using this method, the scientists also investigated the function of one particular point of contact that may be implicated in disease. The results of this study were published in Nature Communications.
The organelles are specialized parts of the cell: The nucleus stores our genetic material; mitochondria produce energy; the endoplasmic reticulum sorts and folds proteins for secretion; the lysosomes degrade proteins; and so on. And each of these is walled off by its own wall or membrane. “But no organelle is an island,” says Staff Scientist Dr. Einat Zalckvar, in the group of Prof. Maya Schuldiner of the Institute’s Molecular Genetics Department. Schuldiner had previously discovered certain contact points and their tethers. But these contacts are so small – just a few tens of nanometers in length – they don’t show up in standard microscope images, and until now only a few of those believed to exist had been uncovered.
Schuldiner and Zalckvar, together with research student Nadav Shai, decided to take a new approach to finding organelle contacts and their tethers. They took yeast cells and coated one of their organelles with one half of a protein that is fluorescent: When whole, this protein glows under the microscope. The other half of the fluorescent protein was attached to different organelles. When the two membranes are in contact, the two halves form a whole, glowing protein that can be detected under any standard fluorescence microscope. Yeast cells are grown in thousands of wells at a time in Schuldiner’s lab and automatically scanned for results, so once their method was perfected, the team could not only test the contacts between a number of different organelles in a single study, but they could also screen all of the yeast genes for potential tethers.
In this way, the researchers were able to map the physical connections between a number of main organelles and to report on four completely new contact sites. “We found that all of the organelles that we tagged are able to physically interact with all of the others,” says Shai. “Tethers organize the cell and assign locations to the organelles. For example, if two organelles must work closely together, they will naturally be tethered together in a certain area of the cell. Tethering also seems to be a way of efficiently transferring, from one organelle to another, certain materials and messages that could do harm in the wrong places,” he adds.
The group then decided to investigate one particular contact site that ties mitochondria to peroxisomes. The mitochondria, as noted, are the energy plants of the cell. Peroxisomes function to break down fats that can be used for energy and to detoxify those that can poteintially harm the cell. For example, the lipids found in the myelin coatings that sheathe the nerve cells are toxic in high amounts; a lack of peroxisome function in those cells leads to their death and to mental retardation. In general, malfunctions in this process can cause several rare but devastating genetic “peroxisome diseases,” including X-ALD, the disease depicted in the movie Lorenzo’s Oil.
A large part of the broken-down fats go directly to mitochondria for energy consumption. In other words, peroxisomes need to connect efficiently to mitochondria. The group succeeded in identifying two genes that produce the mitochondria-peroxisome tethers. Working with a group of peroxisome disease specialists in the Academic Medical Center of the University of Amsterdam, the Netherlands, the team identified the function of this contact site: transferring the broken-down fats to mitochondria for final breakdown. The study suggests that certain peroxisome diseases could actually be tether disorders, that is, a failure of the peroxisome to get fat products to where they are needed.
Tethers organize the cell and assign locations to the organelles
“Yeast cells,” explains Zalckvar, “are nearly the same as ours when it comes to basic functions. So the contacts and tethers we see in yeast probably exist in our cells as well. But yeast are easy to grow, they have fewer genes, and they are well-studied. Once we identify genes in yeast cells, we know where to look in human ones.”
Shai: “Many labs are already adopting our technique to discover new connections within cells. Since it only requires standard microscopes and tools that are found in most labs, and as it works equally well in human cells, we think it will prove useful to many geneticists and cell biologists.”
Zalckvar: “The discovery of contact sites within the cell in recent years has really opened our eyes. If, only a few years ago, we thought that organelles floated within the ‘soup’ of the cell and picked up messages floating in this soup, we now know that each organelle has its own location within the cell, and this location is determined by the network of tethers that also provide a direct means of passing materials through physical contact.” “This understanding of how organelles are organized and interact has revolutionized cell biology,” says Schuldiner. “We believe that our new method of uncovering contacts will lead scientists down new and interesting paths in the years to come.”
Prof. Maya Schuldiner's research is supproted by the Edmond de Rothschild Foundations; the Foundation Adelis; the Berlin Family Foundation; and the University of Michigan. Prof. Schuldiner is the incumbent of the Dr. Gil Omenn and Martha Darling Professorial Chair in Molecular Genetics.