Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
A developing embryo is a site of massive migration: Not only individual cells, but entire groups of cells move about before assuming their assigned spots. Such migrations are largely guided by chemical signals, but scientists have long suspected that the physical structures the developing cells encounter on their way may be no less important in determining their route. Prof. Nir Gov of the Weizmann Institute of Science’s Chemical and Biological Physics Department and researchers from the University of California have shown – in a living organism – that cell clusters integrate both chemical and physical cues in choosing their path.
Understanding how embryonic cells end up where they belong is essential because mistakes in their journey can be fatal to the embryo, or they may lead to birth defects. This includes an entire group of defects affecting the brain and nervous system known as neuronal migration disorders. Understanding how cell clusters move may also shed light on cancer metastasis, as malignant cells sometimes spread in groups.
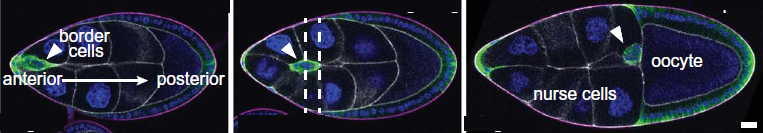
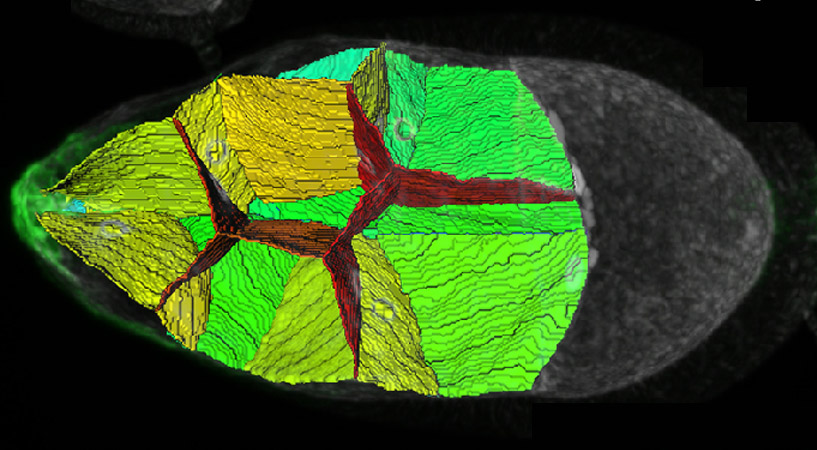
The scientists studied a group of about ten fruit fly cells known as border cells. These are cells that make the move from one side of the fly embryo to all the way to the opposite side in the early stages of development. The cells move in a cluster, following the shortest possible route along the embryo’s central axis. After examining images of fruit fly embryos obtained by the team of Prof. Denise J. Montell at the University of California, Santa Barbara, Gov suggested that apart from chemical signals, the cluster cells are directed along a straight central line by the geometrical placement of supporting cells in the space along the path. They noted numerous groups of three or more cells, each group arranged around a narrow empty space in the middle. Gov thought that these spaces could allow the cluster to squeeze through.
His hunch proved to be right, but for a more complex reason. The mathematical model of cellular movement that he developed with colleagues from the University of California, San Diego, revealed that the empty spaces indeed facilitate the movement of the cluster, but not because they simply enable entire cells to squeeze past. Rather, it turned out that as the cluster moves along, its front cells put out finger-like protrusions. These fingers can squeeze more easily into the spaces at the junctures of cellular groups than in between two adjacent cells that are tightly pressed against one another. And once the fingers are in, the front cells become stabilized, pulling the rest of the cluster behind them.

When Montell and colleagues created mutant fly embryos with different arrangements of supporting cells in the middle, the border cell cluster failed to move in a straight line, despite being guided by the usual chemical signals. These experiments supported the findings of Gov’s model, according to which the geometrical structure of the supporting tissue is crucial to determining the cells’ journey through the embryo.
Study participants included Dr. Wei Dai, Dr. Xiaoran Guo, Dr. James A. Mondo, Dr. Joseph P. Campanale, Dr. Haley Burrous and Dr. Sebastian Streichan of the University of California, Santa Barbara; Dr. Yuansheng Cao and Prof. Wouter Jan Rappel of the University of California, San Diego; and Brandon J. Montell.
Prof. Nir Gov's research is supported by the Ilse Katz Institute for Material Sciences and Magnetic Resonance Research; and the Harold Perlman Family. Prof. Gov is the incumbent of the Lee and William Abramowitz Professorial Chair of Biophysics.