Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
We generally think of mucus as a nuisance, and we become aware of it only when we want to clear our nose or throat. But our bodies produce this sticky fluid in large amounts – more than a liter per day – for a good reason: It protects the inner linings of the body that come into contact with the outside world. In the lungs, microscopic hair-like structures sweep mucus along the airway surface to flush out entrapped particles and bacteria. In the digestive tract, mucus keeps the intestinal contents at a safe distance from the gut’s surface and lubricates its lining, while at the same time housing and feeding healthy gut microbes. Researchers at the Weizmann Institute of Science have revealed the assembly mechanism by which the major component of mucus, a protein called mucin, forms spaghetti-like polymers that contribute to mucus’s gelatinous texture.
When the body fails to produce mucus of the right structure or consistency, or in the right locations, disease can arise. If lung mucus is too thick, it can clog the airways, as occurs in cystic fibrosis; if intestinal mucus is not firm enough, it won’t keep bacteria away from the delicate cells underneath, leading to colitis. In malignancy, mucins produced by cancer cells can interfere with treatment by shielding these cells from chemotherapy. Understanding mucin’s assembly mechanism may prove valuable in helping to repair defective mucus or in selectively breaking down the mucin shield around tumor cells. This understanding can also be used to design artificial mucin-like polymers for various applications in biotechnology – for example, in therapeutic creams and ointments.

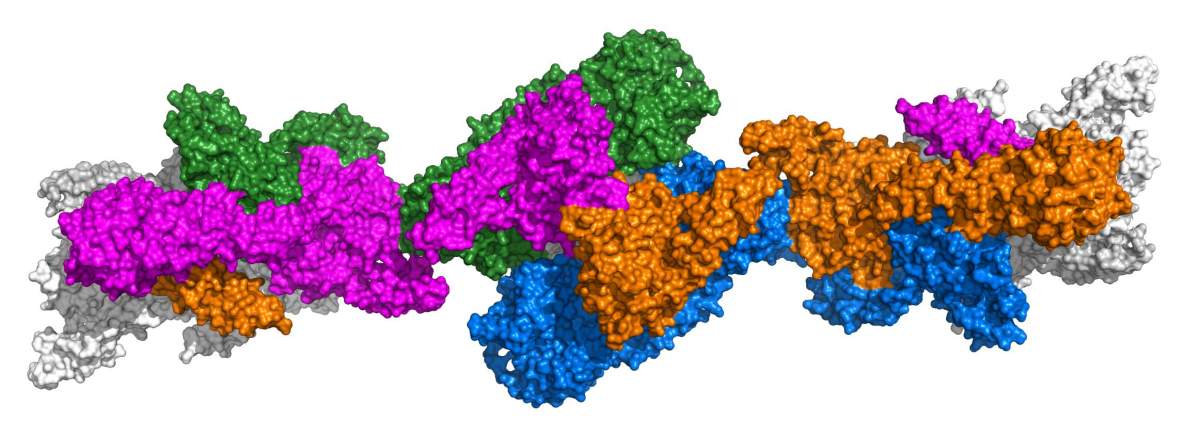
Even though mucins are among the largest proteins in the body – they are composed of more than 5,000 amino acids – they are still not big enough to carry out their biological functions alone. Instead, about 1,500 amino acids at the beginning of mucin proteins (the “head”) and about 1,000 amino acids at the end (the “tail”) participate in the formation of larger structures – by first linking tail-to-tail, and then head-to-head. The several thousand amino acids in the middle of each mucin molecule are flexible and have many sugars attached. The enormous size, flexibility and sugar-coating of mucins, along with their linking tendency, make it difficult to visualize them using standard techniques and understand how they work. It is no wonder then that the way mucin molecules stick together has been a matter of dispute. Furthermore, it was unclear how they formed the spaghetti-like polymers rather than looping into small tortellini-shaped rings.
Research student Gabriel Javitt, who led the study in the lab of Prof. Deborah Fass of the Weizmann Institute’s Structural Biology Department, together with other team members, managed to purify the head region of an intestinal mucin protein and found that this part of mucin assembles into long filaments under slightly acidic conditions. Such conditions indeed prevail in the Golgi organelles of cells, in which mucins are known to assemble. When Javitt and colleagues analyzed the structure of the mucin filaments using the Weizmann Institute's state-of-the-art Titan Krios electron microscope, they unraveled a remarkable, step-wise mechanism by which mucins organize themselves into these complex structures with precision and control.
This process is analogous to sticking little pieces of pasta end-to-end
First, the high-resolution, 3D structure of frozen mucin filaments clearly showed that the protein’s heads link together in pairs and not in groups of three, as some scientists had argued. Moreover, the images, coupled with 3D computer modeling, enabled the scientists to figure out how the looping into tortellini shapes is prevented.
The researchers saw from the structure that the secret lies in the way mucin molecules, already linked into pairs by their tails, are packed inside the Golgi organelles. Had the two heads of a pair stuck to one another, they would have formed a closed loop, but the acidic environment and the heads’ spatial positioning in the filament prevent this from happening, by keeping the sticky parts of the heads belonging to the same pair away from one another. In contrast, the sticky parts of mucin heads from neighboring pairs are held at just the right angle and distance to become pinched together and, once joined, to become stapled together with strong ties called disulfide bonds. This process is analogous to sticking little pieces of pasta end-to-end rather than closing them into rings. Ultimately, dozens of mucin pairs become linked in this manner.
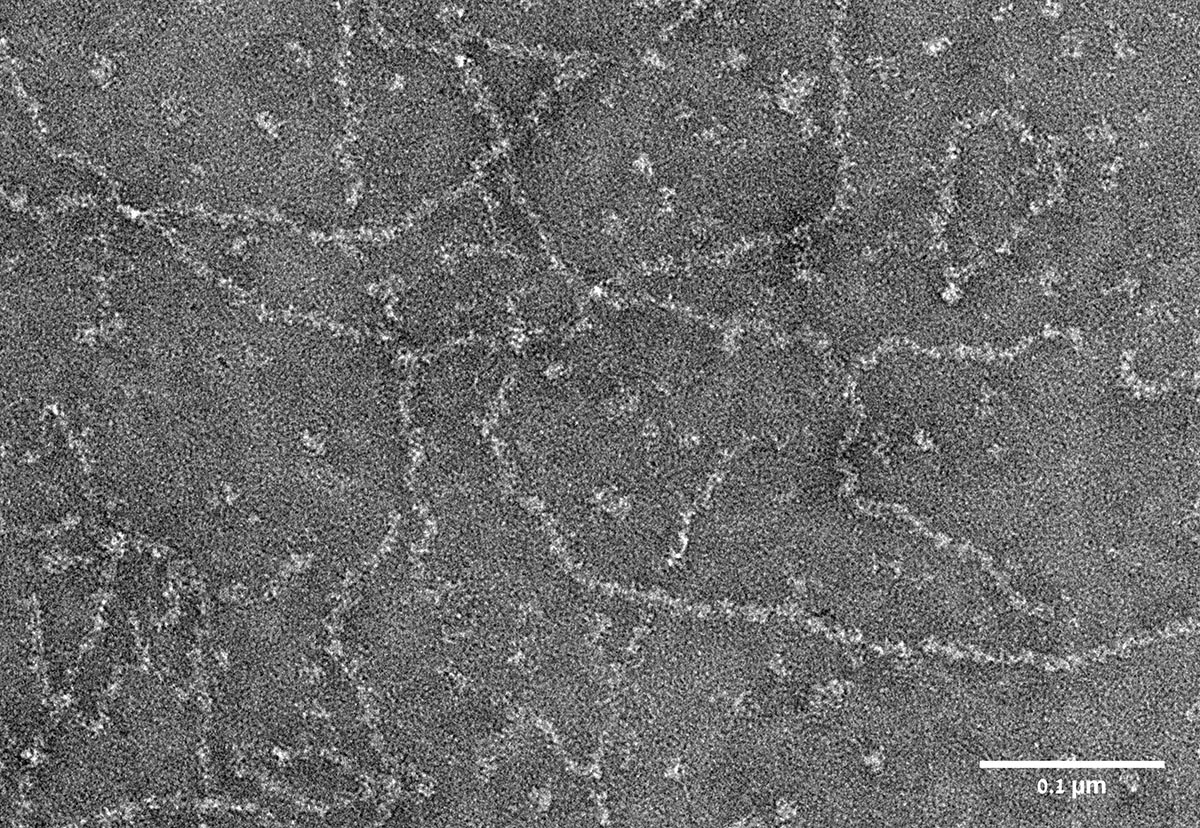
When the assembled mucin polymers are secreted into the intestines or lungs, the filaments stretch out, their compact structure expands, and the mucins absorb a huge amount of water, giving mucus the gooey properties for which it is known. At the same time, the disulfide bonds that were formed inside the cell still hold the mucin chains together, preserving their spaghetti-like structure.
With the knowledge gained from this work, scientists can now imitate the newly revealed mucin assembly mechanism in bioengineering, or they can attempt to interfere with or enhance this mechanism for various therapeutic purposes.
Study participants included Lev Khmelnitsky, Lis Albert, Lavi Shlomo Bigman, Dr. Tal Ilani, Prof. Yaakov Levy and Prof. Ron Diskin from the Structural Biology Department; Dr. Nadav Elad from the Chemical Research Support Department; and Dr. David Morgenstern from the Nancy and Stephen Grand Israel National Center for Personalized Medicine.