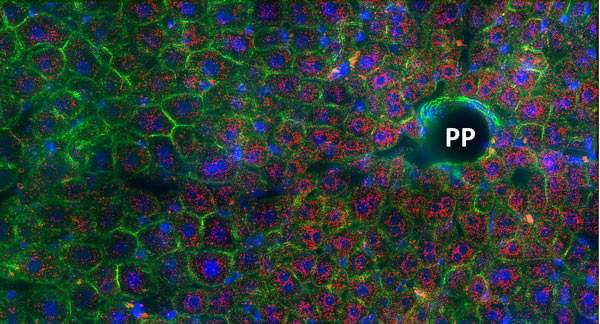
This flexibility can be crucial in performing the liver’s dynamic functions – for example, in regulating blood glucose. As part of ongoing maintenance, the liver takes up extra glucose when its levels are too high, then gradually releases it or synthesizes new glucose when levels drop. As glucose levels rise within minutes after a meal, the synthesis of new glucose must be able to stop instantly. The short-lived mRNA, which quickly disappears from the cell once gene activity is shut down, is perfectly suited to this end.
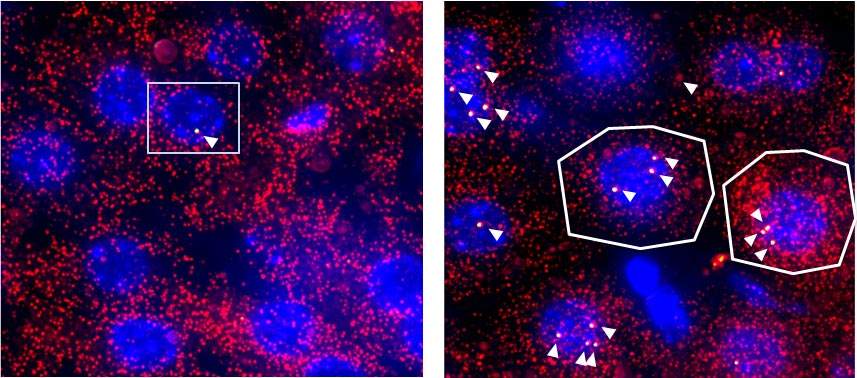
Indeed, the Weizmann study found that the mRNAs of two genes essential for glucose production are extremely short-lived. To compensate for their brief life span, they are produced in longer than average bursts, presumably to reduce the variability among cells caused by the bursts. On the other hand, other mRNAs, with a longer life span, are produced in shorter bursts.
The scientists believe the bursty expression of genes could have evolved because it can protect the DNA from damage: Genes are physically more exposed when active, so by being active only at intervals, rather than permanently, they are less vulnerable to surrounding toxins. This feature is particularly important in an organ like the liver, which is involved in filtering out harmful substances.
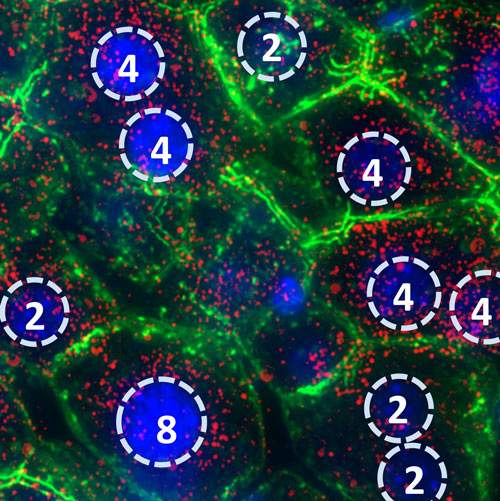
The scientists also believe that the bursty activity may help explain a baffling feature of many liver cells: the presence of multiple copies of the genome, comprising four or eight DNA strands instead of the usual two. Their proposed explanation goes as follows: The bursts cause mRNA levels to fluctuate at random, but thanks to the extra DNA copies, each of which produces mRNA, this randomness is averaged out among cells. As a result, different liver cells end up producing a particular mRNA in a uniform manner. Indeed, in a “factory” such as the liver, where cells work together towards a common physiological goal, excessive variability among cells caused by such bursts could have been a disadvantage.
The team that performed this research included Dr. Keren Bahar Halpern, Sivan Tanami, Shanie Landen, Michal Chapal, Liran Szlak, Anat Hutzler and Anna Nizhberg.
Further clarification of the bursty gene expression in the liver may help reveal such mechanisms of faulty liver function as defective glucose metabolism, which leads to diabetes. The scientists have also found indications that bursty gene expression may be found in organs other than the liver, a finding that opens new ways of investigating the control of gene activity in different tissues.