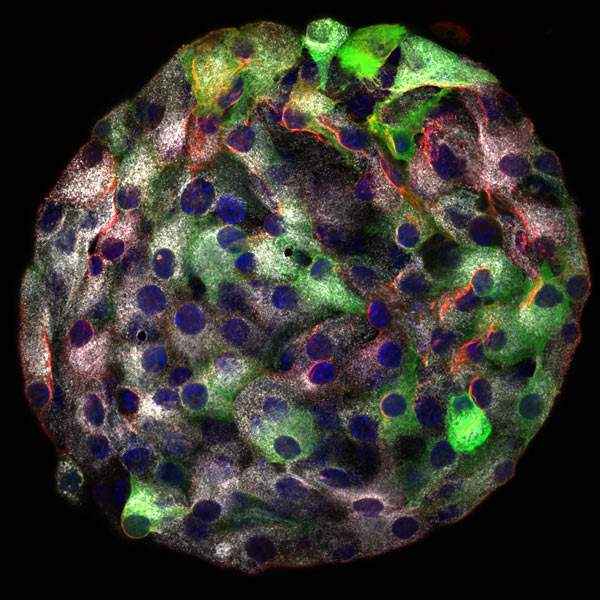
Within the pancreas are little spherical structures called islets of Langerhans – a name that might conjure up images of cells lazing on a remote beach, sipping tropical drinks. That picture could not be further from the truth: The so-called beta cells, the major component of these islets, are some of the busiest, most connected cells in our bodies. They do sip, but it is the sugar in our bloodstream they taste; they then secrete insulin back into the bloodstream to regulate the metabolism of that sugar. And, according to new Weizmann Institute research, these cells submit to a very tight organization. The study, which appeared in Cell Reports, revealed, for the first time, an image of beta cells that are “edgy.” The details of how beta cells are shaped may change our understanding of how they function and lead to new insights into how glucose is regulated in the body.
Beta cells are those affected by diabetes: In type 1 diabetes they are attacked by an autoimmune response in the body, and in type 2 diabetes the body cells do not respond to the insulin they secrete, which eventually leads to their dysfunction. The beta cells are a minority in the pancreas, but the island-like arrangement within the “sea” of pancreatic cells enables them to function as an organ within an organ. A closer look at the arrangement of cells in an islet of Langerhans shows that they assemble in “rosette” patterns around veins running through the pancreas, with small arteries encircling the rosettes.
Prof. Ben-Zion Shilo, research student Erez Geron and Dr. Eyal Schejter of the Molecular Genetics Department wanted to zoom in a bit closer – to the molecular arrangements of these cells. Many had assumed that the cells were polar – that they concentrate some functions on one side and others on the opposite side. Polarity, according to this view, would enable the beta cell to compartmentalize its activities, possibly sensing on one side and secreting on the other. In addition, beta cells are related to other polar cells – for example, those that line the intestine. But no one had actually managed to observe clear hallmarks of polarity in beta cells.
To investigate, Geron studied proteins on the cells’ outer membranes – where sensing and secretion take place. Isolating individual islets of Langerhans from a mouse pancreas, he inserted a fluorescent protein into several of their cells. The team then devised a technique that allowed them to visualize filamentous actin, a major component of the cellular cytoskeleton, just within a few individual cells of each islet, thus providing good contrast right at the cells’ contours. Since islets of Langerhans can be kept alive in the lab dish, the researchers were able to observe the shapes of beta cells at work.
Cell design on a line
What they saw surprised them: Rather than a polar arrangement, the actin in the membrane formed stiff, linear edges, like those on a cube, along the length of the cell, giving it the appearance of an angular tent with poles. But these lines were more than just support poles: Numerous thin protrusions extended from the cell along these lines, and membrane features congregated along them – signaling proteins, channels for letting in glucose, others for trading short messages encoded in calcium ions with neighboring cells, and even outlets for secreting insulin.
Why do these particular cells reject their polar heritage and go for edgy shapes? Why would they take all of their functions – sensing glucose, releasing insulin and intercellular communication – and align them together along these cell edges? Shilo says that they do not yet have all the answers, but he and his group have some definite clues. For example, they think that keeping the sensing and secreting machinery close together could increase efficiency and/or cut down on response time.
Further research will be needed to understand the exact function of the edges, but Shilo says the study points to an important role in cell-to-cell communication. Indeed, one of the major implications of these findings is that, when it comes to cell design, relations between neighboring beta cells are critical. It is communication between cells that creates the tent-like shape in the first place: Beta cells that are cut off from the others soon lose their edges. And if the “tent poles” help facilitate communication, then the protrusions along their length are likely to play a role in passing messages back and forth, possibly by helping align the membranes’ pores, which are just big enough to admit the calcium ions. “These findings suggest a mechanism for beta cells to function in unison,” says Shilo. “Coordinating their actions might help prevent random responses by single cells.”
Although the work was done on mouse pancreatic cells, there is already some evidence that human beta cells also have edges. These findings, says Shilo, shed light on the ways that insulin-producing beta cells function, and they could lead to new ways of thinking about the normal complex, coordinated activity of these cells, which play such a central role in our health.
Prof. Ben- Zion Shilo's research is supported by the M.D. Moross Institute for Cancer Research; the Carolito Stiftung; and the Mary Ralph Designated Philanthropic Fund. Prof. Shilo is the incumbent of the Hilda and Cecil Lewis Professorial Chair of Molecular Genetics.