Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:
Connections are crucial. Bacteria may be most dangerous when they connect -- banding together to build fortress-like structures known as biofilms that afford them resistance to antibiotics. But a biomolecular scientist in Israel and a microbiologist in California have forged their own connections that could lead to new protocols for laying siege to biofilm-protected colonies. Their research was published in the Proceedings of the National Academy of Sciences (PNAS), USA.
This interdisciplinary collaboration began with a lecture given at the Weizmann Institute of Science in the Life Sciences Colloquium. Prof. Dianne Newman of the California Institute of Technology was the speaker, and the Institute’s Prof. Sarel Fleishman, of the Biomolecular Sciences Department, decided to attend, even though the lecture had no immediately apparent bearing on his own research. Newman described an enzyme she had discovered that could interrupt the metabolism of the biofilm-building bacteria, Pseudomonas aeruginosa. The enzyme interferes with the functioning of a molecule (pyocyanin) that is generated by the bacteria as they reach a high cell density and start to run out of oxygen, and it is thus responsible for helping bacteria deep within the biofilm remain viable as well as better tolerate conventional antibiotics. This molecule, however, is a double-edged sword: It can also be toxic to P. aeruginosa in the outer layers of the biofilm, where oxygen is present. Since pyocyanin impacts both biofilm development and antibiotic tolerance, Newman’s lab focused on identifying ways to disrupt its activities. Newman's only problem, she said, was that the newly discovered pyocyanin-blocking enzyme was unstable and produced in minute amounts, and thus far, standard lab methods for growing such proteins had not been successful.
Pseudomonas aeruginosa is an opportunistic bacterium that causes disease mainly in those with underlying conditions: in the lungs of cystic fibrosis patients, the peripheral wounds in diabetics and on various implanted medical devices of hospital patients. Hard-to-eradicate biofilms may help infections return even after treatment, contributing to the bacteria’s growing antibiotic resistance, particularly in hospital-acquired strains.
After the lecture, Fleishman suggested to Newman that they try a new approach to producing larger quantities of the enzyme. His lab specializes in computational protein design, and some of their recent work had involved redesigning vaccine proteins to make them more stable.
Rosalie Lipsh-Sokolik, a research student in his lab, together with Dr. Olga Khersonsky, a research associate, took up the challenge of designing an improved, more stable biofilm-busting enzyme. But the enzyme was unlike any Fleishman’s lab had worked with before, and it would require them to develop a new methodology: It was a trimer – three identical copies of a protein bundled “like barrels strapped together,” says Fleishman, and that meant that, in addition to the structure of the individual protein, they would need to understand how the entire package fit together.
The group’s first step was to map the enzyme down to its atomic structure. This gave them a detailed picture of the forces that hold the protein together. When they added the resulting models of the three copies together to understand the trimer formation, they noticed that the areas of contact between copies were poorly packed, atomically speaking, and they thought these particular weak points would be a good place to start in designing a more stable structure.
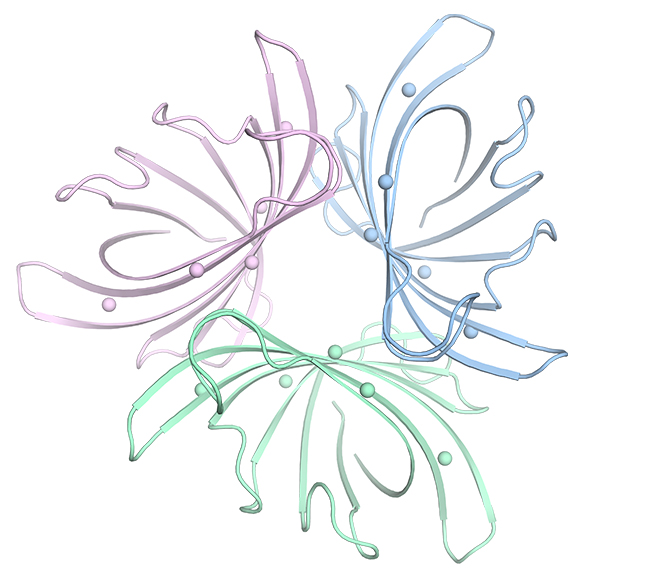
But even after narrowing down the potential sites for adjustment, the number of design possibilities for such a protein complex was huge. Lipsh-Sokolik ended up adopting a combined, two-pronged approach. The first was to look for proteins made by other bacteria that are similar but slightly different, to see what could be borrowed. The second was a sort of “subtle” atomistic design approach, identifying just a dozen or so points on the enzyme that might be tweaked and trying out different modeled combinations of amino acids at just those points.
The beauty of the computer design methods developed in Fleishman’s lab is not only the fact that they can produce, in a very short time, hundreds of thousands of different possible protein designs, but they also rank them from most-likely to work to not likely to work at all. Still, the only way to know if your hypothesis is correct -- about the areas that need reinforcement or the ability of an enzyme to still function despite changes to its protein sequence -- is to make those proteins and test them in real biological systems. Enter Dr. Chelsey M. VanDrisse, a postdoctoral fellow in the Newman lab, who led all the experimental tests of the Fleishman lab designs.
Eight of the ten designed enzymes were produced in larger-than-normal quantities without compromising their biofilm-fighting abilities
Fleishman admits his team was nervous when VanDrisse and Newman told them that they could conduct experiments on only ten of the designed trimer enzymes due to the highly challenging nature of the experiments. Their challenges were not limited to creating these new proteins, but included figuring out how to purify them in sufficient quantities in the lab and then testing them on real biofilm-building Pseudomonas aeruginosa in combination with standard antibiotic treatment. The question was, could the team not only produce a more active protein, but determine whether its application could facilitate biofilm control and begin to understand the mechanisms underpinning the enzyme`s effects?
"Both teams were over the moon when the results came in," says Fleishman: Eight of the ten designed enzymes were produced in larger-than-normal quantities in Newman’s lab, without, it seemed, compromising their biofilm-fighting abilities. VanDrisse jokingly raved that she could now produce so much protein she “could have put it in my cereal every morning!” “This showed that our hypothesis about the contact areas was correct,” says Fleishman. One enzyme seemed especially robust and was produced in substantial amounts, so VanDrisse and Newman went all the way: They set out to check if this version of the enzyme could, at least in the lab, work together with a commonly used clinical antibiotic to eradicate the biofilm.
In fact, they found the enzyme, in combination with this antibiotic, worked much better than they had expected. Further analysis suggested the enzyme first helps the antibiotic kill the bacteria in the oxygenated outer regions of the biofilm in a way that had not before been seen, leading, in short time, to a significant reduction in the total number of viable biofilm cells.
Fleishman adds that, as the collaboration between two groups of scientists who normally read different journals, attend different conferences and experiment with very different methods on different scales deepened – VanDrisse even making it to the Weizmann lab just before the first COVID-19 lockdowns – he realized that what had started for him as a test of his lab’s computational protein design methods now had a very real chance of leading to a cure for some of the most aggressive bacterial infections. It all came down to making the right connections.