The molecular machines that defend our body against infection don’t huff and puff, but some of them apparently operate on much the same principle as a steam engine. Weizmann Institute scientists have discovered a mechanism that controls inflammation in a way that resembles a steam-engine valve: Just when the inflammatory mechanism that protects cells against viruses reaches its peak of activity, the molecular “steam-release valve” intervenes, restoring the mechanism to its resting state, ready for re-activation.
This finding might shed new light on such inflammatory disorders as rheumatoid arthritis or inflammatory bowel disease, and point the way to developing effective therapies.
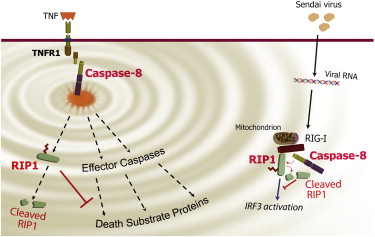
How does the cellular “steam-release valve” work? The scientists have discovered that its crucial component is the enzyme called caspase-8. When the cell is invaded by a virus, caspase-8 joins a large molecular complex that forms in order to send out an inflammatory signal. However, this same signal, once triggered, makes sure that the inflammatory response will eventually be shut down. The mechanism can be likened to the peak of the steam cycle, when the valve opens, releasing steam and restoring the engine to its initial state. In the case of the cell, the inflammatory signal prompts caspase-8 to destroy a protein called RIP1 – a crucial signal amplifier – after RIP1 has reached a state in which it can produce maximal amplification. The inflammatory cycle is thus completed: The signaling mechanism, precisely after reaching its peak activity level, returns to its neutral state, ready to enter yet another inflammatory cycle should the viral attack on the cell continue.
Until recently, caspase-8, discovered by study leader
Prof. David Wallach of Weizmann’s Biological Chemistry Department some 15 years ago, was known to prevent inflammation in only one way – by causing damaged cells to self-destruct in a process called apoptosis. During this process, the contents of the dying cells are prevented from spreading around and triggering inflammation. The present study, reported recently in
Immunity, has revealed an entirely new mechanism by which caspase-8 can control inflammation more directly. The research was performed in Wallach’s lab by Drs. Akhil Rajput, Andrew Kovalenko, Konstantin Bogdanov, Tae-Bong Kang, Jin-Chul Kim and Jianfang Du, together with Seung-Hoon Yang.
The study results might be relevant for various types of inflammation – not only those caused by viruses – and could thus provide important insights into inflammatory disorders. Since such disorders may occur when the inflammatory response fails to be shut down properly, it’s possible that caspase-8 malfunction and the resulting excessive activity of the RIP1 “signal amplifier” might be involved. And if this is indeed the case, a new treatment approach could aim at blocking RIP1, thereby fighting inflammation in a selective, precise manner.
Prof. David Wallach’s research is supported by the M.D. Moross Institute for Cancer Research; the Shapell Family Center for Genetic Disorders Research; and the Leona M. and Harry B. Helmsley Charitable Trust. Prof. Wallach is the incumbent of the Joseph and Bessie Feinberg Professorial Chair.