Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:

Our views on the contribution of cell death to immune defense might need to be revised: Such death might occur in different situations than currently believed. This conclusion emerges from an opinion piece in the journal Immunity by Prof. David Wallach of the Weizmann Institute of Science’s Biomolecular Sciences Department. In calling on scientists to rethink programmed cell death, Wallach relies on the research of numerous laboratories, including more than three decades of his own studies. In the 1980s, he was among the first to isolate a natural molecule called tumor necrosis factor, or TNF, which causes the death of cells and plays a crucial role in immune defenses but which can also promote uncontrolled inflammation. Wallach discovered one of the main molecular mechanisms – the extrinsic pathway – by which a program of cell death called apoptosis can be activated by TNF, among other factors, and he identified two TNF-blocking molecules that led to the development of widely used drugs for such chronic inflammatory diseases as rheumatoid arthritis, psoriasis and inflammatory bowel diseases. Currently, Wallach is among the leading scientists to study a protein called MLKL, which plays the key role in yet another form of programmed cell death – necroptosis. In March 2018, Wallach was awarded the prestigious Paul Ehrlich and Ludwig Darmstaedter Prize for his pioneering studies.
Q. Why do you believe the occurrence of programmed cell death in immune defense needs to be reevaluated?
DW: Our current knowledge of programmed cell death is largely based on a source of information that is far from optimal and might therefore yield an inaccurate picture. Since cells that die in a programmed manner are rapidly eliminated from the body, detailed documentation of the occurrence of such death within the mammalian body is hard to obtain. Therefore, most of our knowledge about programmed cell death is based on studies outside the body – ex vivo – yet these studies don’t provide an accurate account of the roles this death plays in the living body – in vivo.
Q. Can you please elaborate?
DW: When occurring in vivo, each program of cell death constitutes part of a broader program, in which death is often the least favored option. In my opinion-piece in Immunity, I provide two sets of examples. One deals with the role of apoptosis in the development of immune cells called lymphocytes; the other focuses on conclusions arising from our studies of necroptosis – a form of death in which the membranes of cells rupture, so it can be accompanied by inflammation. This subject has been addressed extensively in my laboratory in recent years, particularly by three students from Korea: Tae-Bong Kang, Seongmin Yoon and Ju-Seong Jeong.
The body operates like the legal system of a country that maintains the death penalty...instituting capital punishment only when all other means have failed
When scientists see inflammation and detect molecules involved in necroptosis, they assume that necroptotic death has indeed occurred, and that it is the cause of the observed inflammation. But findings in my own lab and in others indicate that this might not always be true. The mechanisms of programmed cell death serve immune defense in a measured manner. The same molecules that trigger necroptosis can also initiate inflammation without inflicting death upon the cells. Cell death is only their last resort. In this sense, the body operates like the legal system of a country that maintains the death penalty. To say that this system aims only at killing people would be wrong, because it is also built on milder punishments and means of rehabilitation, instituting capital punishment only when all other means have failed. Similarly, within the body, cell death is not a goal in itself but part of a comprehensive “correctional” program.
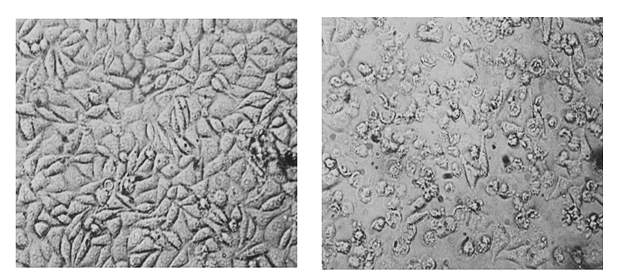
Q. What findings have led to this conclusion?
DW: Many studies, including those in my own lab, have shown that proteins triggering or inhibiting cell death often “moonlight” by performing additional jobs in the cell. TNF, for example, was originally known only for causing death in cells, but my lab found that this wasn’t the case – we were the first to show that TNF also serves other functions. Another example is a protein called caspase-8, discovered in my lab. We initially found that it initiates apoptosis. However, my laboratory and other scientists later showed that it also controls various other functions – for example, it blocks the induction of necroptosis. Eliminating the latter function of caspase-8 triggers both inflammation and necroptosis. We, however, found that caspase-8 can also control inflammation more directly, not via necroptosis. Therefore, when cells are infected by viruses or bacteria that block caspase-8, the infection can trigger inflammation without killing the infected cells.
More recently, we’ve discovered moonlighting in yet another molecule, the protein MLKL that is involved in necroptosis. MLKL kills cells by making holes in their membranes; but we have shown that it also performs non-deadly tasks in the cell, and that in this way it can trigger inflammation without causing cell death.
Q. How can you prove that the same molecules can cause inflammation with and without cell death?
DW: Studies in mice show that when the enzyme I mentioned earlier, caspase-8, is removed from tissues, this removal leads to severe inflammation. Scientists initially assumed that this inflammation is always caused by a resultant necroptosis, in which the contents of the cell spill out. But a study published by my lab a few months ago showed that even if we remove the molecular machinery responsible for necroptosis in these mice – in particular, an enzyme called RIPK3 – they still develop a certain degree of inflammation.
We need to think of better tools to properly assess the role of cell death in a variety of situations
Q. So finding lethal tools doesn’t necessarily mean a crime has been committed?
DW: Exactly. If we find knives and blood stains at a particular site, we can’t tell if this is a crime scene, or perhaps a place in which a life-saving operation has been attempted. After all, the body triggers inflammation in an attempt to prevent damage, for example, the spread of infection.
Q. How can scientists achieve a better assessment of the role of programmed cell death?
DW: First of all, it’s essential to become aware of the problem – that our current view of the situations in which such death occurs in the body is based on insufficient evidence. Once we appreciate this, we need to think of better tools to properly assess the role of cell death in a variety of situations in vivo. For example, we need to find ways of withholding the clearance of dying cells in experiments done in vivo, so that the occurrence of death can be properly monitored, and to improve the imaging of the death itself and of the molecules that take part in different death programs.
Q. Why is it important to know whether programmed cell death has occurred?
DW: First of all, obtaining a correct view of what happens in the body is what biology is all about. Moreover, cell death is believed to contribute to immune processes taking part in a variety of diseases that affect billions of people. A more accurate view of the connection between cell death and these processes may lead to improved therapies. For example, necroptosis that results in inflammation occurs in stroke, heart attack, viral infections and numerous other conditions. But the mechanisms responsible for necroptosis also induce inflammation without cell death in these diseases. Our approach to developing new drugs would be different in the two scenarios – with and without cell death. If the inflammation is caused by cell death, then we need to block the death, but if not, the drugs will need to target the non-deadly functions of the molecules involved in necroptosis.