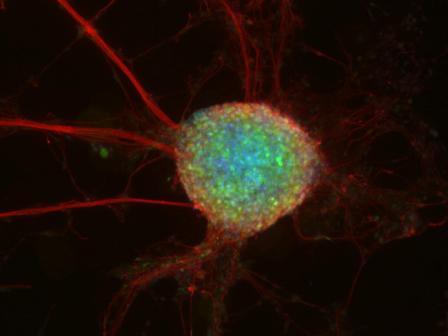
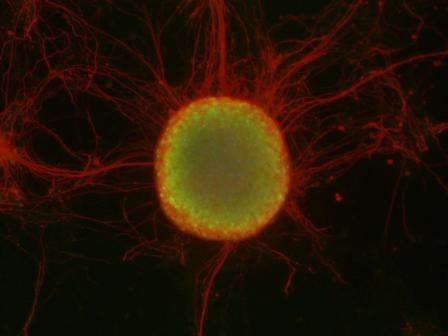
The potential of embryonic stem cells is almost unimaginably huge. From one undifferentiated cell, an entire organism composed of diverse and widely varied tissues and organs is created. Now, scientists are looking to expand the potential of these cells: As medical tools, they might be used to restore injured cells and tissues, and help to treat such genetic conditions as autoimmune diseases and degenerative nervous system disorders. “The main hurdle that stands in the way of medical embryonic stem cell application today is getting the stem cells to differentiate properly. We still don’t know how to reproduce that process efficiently, precisely and in quantity,” says Dr. Jacob (Yaqub) Hanna, who recently joined the Institute’s Molecular Genetics Department. In his new Institute lab, he and his research team will try to answer basic questions on the mechanics of stem cell differentiation and development, in hopes of advancing the use of stem cells in medicine.
Sending cells back in time
Another area of stem cell research that interests Hanna is a recent breakthrough: Several years ago, scientists demonstrated that mature, adult skin cells could be “sent back in time,” to return to their embryonic stem cell state. Surprisingly, this reprogramming was accomplished with the insertion of only four genes into the adult cell genome. Creating stem cells from adult cells solves various ethical problems inherent in using embryonic stem cells from human eggs or embryos. But there could be other advantages, as well. Patients with certain diseases could be treated with their own cells, avoiding the need for matching donors. Cells taken from a person with a genetic disease could be reprogrammed into stem cells; genetic engineering techniques would then be used to repair the genetic cause of the disease, and the renovated stem cells directed to undergo the required differentiation before being reimplanted in the patient.
In his postdoctoral research, Hanna and his lab partners were the first to prove the feasibility of this method, showing it could cure sickle cell anemia in mice. In another investigation – into why reprogramming is successful in only a small percentage of adult cells – he found a number of “main switches” all of which must be turned on for the process to complete itself. To gain a better understanding of the mechanisms involved in transforming a cell from one type to another, Hanna plans to uncover the exact functions of these switches and find out why they are so important. Another approach, says Hanna, will be to investigate how an egg can easily do what scientists struggle to achieve: In the lab, it can reprogram adult cells to generate perfect embryonic stem cells.
In addition to the goal of unraveling the basic mechanisms of differentiation and development, and his longer-term aims in the field of regenerative medicine, Hanna says that stem cell research can supply new, sorely needed tools for research into human gene-based diseases, among them Type 1 diabetes, Parkinson’s and Alzheimer’s. These diseases involve degeneration of the cells, making them hard to study. But Hanna plans to create stem cells from the adult cells of patients. He can then apply such genetic engineering methods as inserting or removing genes, or adding markers to create useful models for researching these diseases – hopefully opening up new pathways to developing treatments.
Award winning researcher
Dr. Jacob Hanna was born and raised in Rama, in the Galilee, into a family of doctors: His grandfather, father and three sisters are doctors. In keeping with the family tradition, he studied medicine at the Hebrew University of Jerusalem, but found he was attracted to research, preferring hours in the lab to treating patients. “My uncle, a scientist who heads a pharmaceutical firm, was my influence and inspiration. This uncle was behind the development of the first antibody approved for the treatment of lymphoma.” In 2007, Hanna completed a combined MD/PhD program, magna cum laude. His postdoctoral research was carried out with Prof. Rudolf Jaenisch of the Whitehead Institute for Biomedical Research at the Massachusetts Institute of Technology (MIT). His research there won him a number of awards; among them, in 2010 he was named one of the 35 innovators under 35 by Technology Review. In 2011, he joined the Molecular Genetics faculty of the Weizmann Institute.
Hanna lives in Tel Aviv. Most of his free time is spent on his favorite hobby – scientific research. But in addition, he manages a Tel Aviv bar together with three good friends.
Dr. Jacob Hanna's research is supported by the Sir Charles Clore Research Prize; the Leona M. and Harry B. Helmsley Charitable Trust; Pascal and Ilana Mantoux, France/Israel; and the European Research Council.