Our bodies are alive – not just with muscles and nerves but with populations of bacteria and other single-celled organisms that are along for the ride. An estimated 1016 single-celled organisms – ten to a hundred times more than our own body’s cells – reside in each and every one of us. In recent years, we have been coming to understand the significance of these microorganisms – both for the healthy functioning of our bodies as well as for the role they play in such diseases as diabetes, obesity and cancer. If researchers once saw these organisms as “stowaways” that had a limited effect, mainly on the digestive system, today they are understood to be an integral part of who we are. Some even relate to the multicellular host and its population of single-celled organisms as a “super organism” with two genomes, the “second genome” being the combined genetic material of the microorganism population, which is a hundred to a thousand times larger than the “first genome.”
Dr. Eran Elinav, a medical doctor and researcher who recently joined the Institute’s Immunology Department, researches the complicated relationship between the host’s body and the population of microorganisms that call it home. In a series of papers published during his postdoctoral research, Elinav discovered a means of communication for passing messages between different cell types, in the process revealing clues as to how the single-celled population contributes to some of the most common diseases in the Western world: diabetes, cancer, arteriosclerosis and inflammatory bowel disease (IBD).
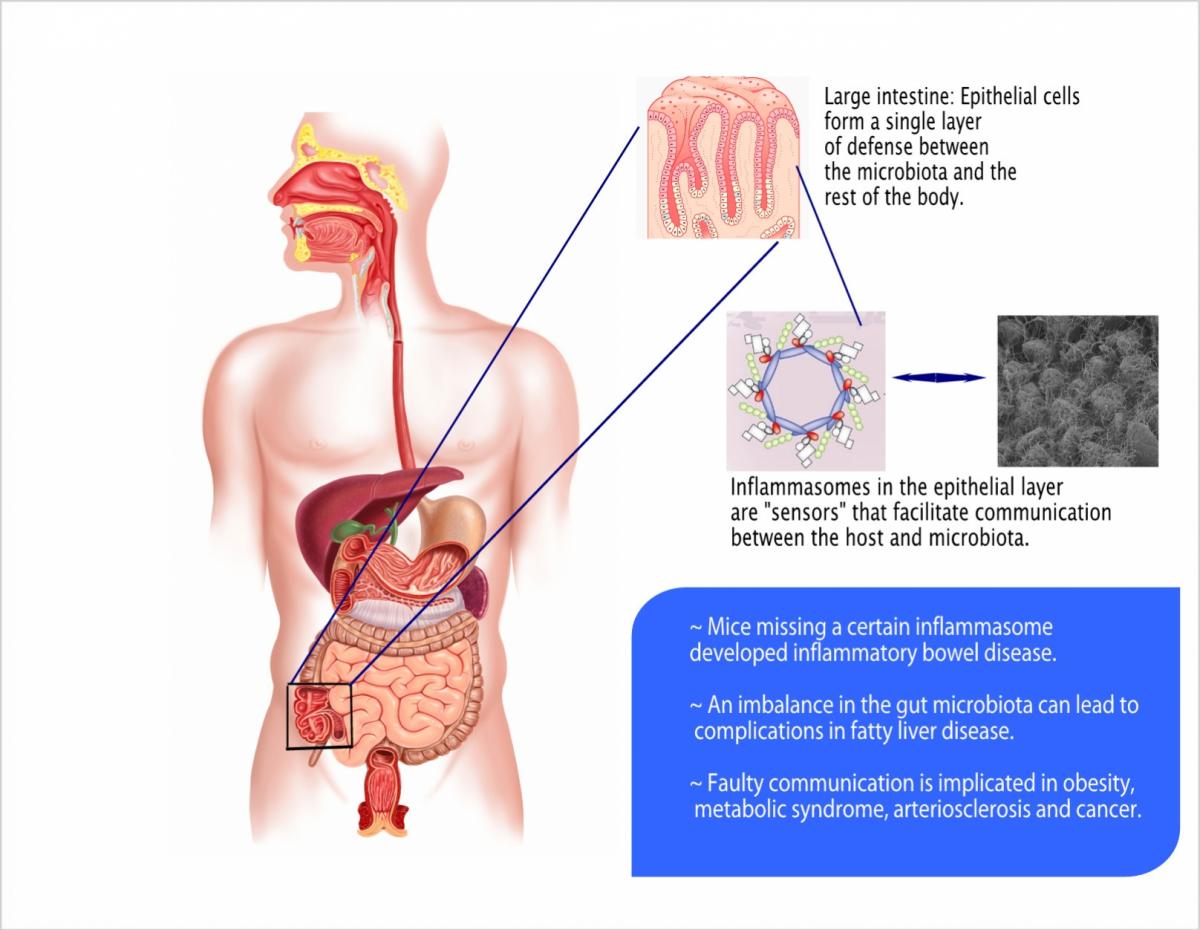
The intimate relationship between microorganisms and their host transpires in a number of places, including the skin and such mucus membranes as the mouth and airways. The most dramatic encounters take place in the digestive system; the population density in the gut is one of the highest on the planet. And yet only the single layer of cells lining the gut – the epithelial cells – keeps the inner parts of our body free from this immense microbial population.
In a study that appeared in
Cell, Elinav found that this layer, which serves as the gut’s first line of defense against unwanted invaders, contains a sort of “snooping device” that helps the immune system keep tabs on the nearby bacteria. The sensor he discovered, called an inflammasome, is one of a group of factors identified in recent years that enable host and bacterial cells to communicate. (Related sensing systems – toll-like receptors – discovered in 1996, were the subject of the 2011 Nobel Prize in Medicine.) When Elinav disrupted the function of this inflammasome in mice, they developed inflammatory bowel disease, thus demonstrating the importance of this factor in keeping the gut healthy.
In further research, Elinav found that this inflammasome may also be involved in metabolic disease and cancer.
In one study, which appeared in
Nature, he created mice lacking the inflammasome in order to investigate a question about the progression of fatty liver disease: Why does this disorder, which affects up to 30% of the human population in the developed world, cause severe problems in only a minority of them? Most people with the disease do not suffer any serious problems, but around 20-30% progress to complications that include chronic liver inflammation, cirrhosis, tumors and even death. Elinav showed that a change in the composition of single-celled organisms in the gut can act as a “switch” that turns on the complications, and that such changes in the bacterial population may also be implicated in diabetes and obesity. Amazingly, in these instances, metabolic disease and obesity may be transferrable between individuals just by a transfer of these microbes between them. In additional research, he revealed a connection between these microbial changes and the propensity to develop cancer.
In his new lab at the Weizmann Institute, Elinav plans to continue this line of research, both in mouse models and in humans. Because this young field presents a variety of challenges, he has brought together in his group people with experience in different areas: immunology, microbiology, metabolics and bioinformatics; and he has established collaborative partnerships with research groups within the Institute and around the world. He also works closely with the Israel National Center for Personalized Medicine, on the Weizmann campus. “Everyone carries around their unique, personal population of microorganisms, and this, in turn, is affected by everything from diet to geography to environmental exposure. Revealing a person’s ‘second genome’ will give us precise tools, for example, to predict how certain diseases will develop or progress, as well as the ability to tailor individual treatments for such diseases as diabetes, atherosclerosis and cancer,” he says.
Pursuing Medicine and Research
Dr. Eran Elinav studied medicine at the Hebrew University of Jerusalem Medical School because, he says: “It is a fascinating profession that combines intellectual and emotional abilities.” During his residency at Hadassah Hospital, he began to take an interest in research. After finishing his residency, in parallel with his work as a doctor in the gastroenterology institute of Sourasky Medical Center in Tel Aviv, he undertook doctoral studies in the group of Prof. Zelig Eshhar at the Weizmann Institute. In 2012, after completing postdoctoral research in the group of Prof. Richard Flavell of the Yale School of Medicine, he joined the Immunology Department of the Weizmann Institute.
Eran is married to Hila, a medical doctor who directs the AIDS center at Hadassah-Hebrew University Medical Center. The couple has twins – Shira and Omri – age 13, and an 8-year-old daughter, Inbal. When he has free time, Eran enjoys water sports, biking and hiking.
Dr. Eran Elinav's research is supported by the Abisch Frenkel Foundation for the Promotion of Life Sciences; the Gurwin Family Fund for Scientific Research; the Leona M. and Harry B. Helmsley Charitable Trust; Yael and Rami Ungar, Israel; the estate of Jack Gitlitz; and the estate of Lydia Hershkovich.


