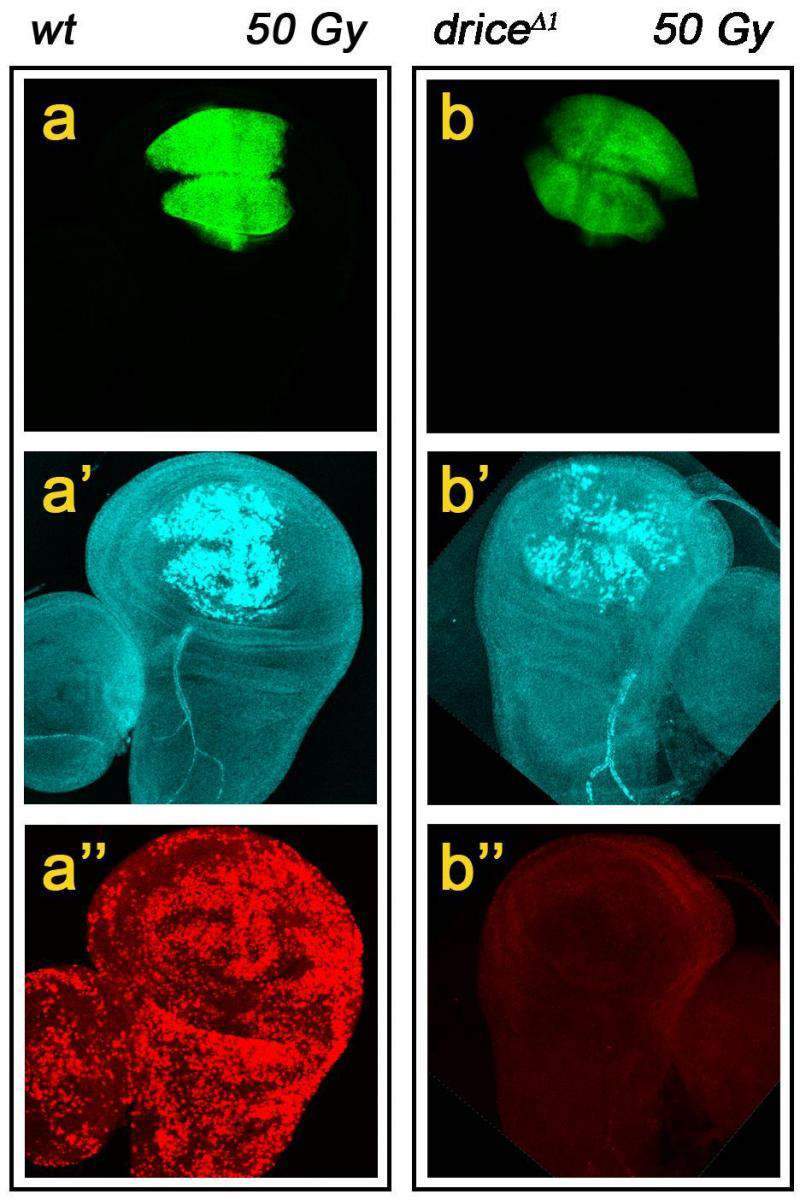
Their results suggest that Drice is the major killing machine: It can destroy a cell on its own, even though it does work more efficiently in the presence of Dcp-1. As for Dcp-1, its major role is to set the pace of the killing, determining how fast and how many cells will die. Dcp-1 can also finish off the cell on its own, but this will happen only if it is present in large amounts.
In fact, one of the study’s central findings is that both caspases can kill cells only when their activity exceeds a certain threshold. Below that threshold, the cell can survive, replenishing the proteins destroyed by the caspases. This finding also explains how certain cells manage to use enzymes such as caspases to promote vital cellular processes without bringing destruction upon themselves: With low or transient levels, the cells avoid apoptosis while employing the caspases for other purposes.
Why are two caspases necessary to perform essentially the same function? The scientists believe that the availability of two “weapons” offers greater precision in controlling the process than would be provided by a single killing enzyme. Drice is more of a sledgehammer, suitable for coarse demolition jobs, whereas Dcp-1 is more of a chisel, best suited to fine-tuned cellular elimination.
The study has shown that the tendency to undergo apoptosis, which varies greatly among different types of cells, is determined by the amount of the caspases present in the cell. Cells less prone to apoptosis are likely to have lower levels of the destructive enzymes. Among these are the brain’s neurons, which are limited in number, and the sperm and eye cells, which would perish upon each contact with the environment if they weren’t so resistant to apoptosis. In contrast, cells that are relatively expendable because they regenerate are more prone to apoptosis and these should have more caspases. These include hair cells and those in the inner lining of the gut, which must be continuously replaced after being damaged by digestive acids.
Florentin and Arama were able to discover these details about the workings of the caspases because they observed their function in living animals – genetically engineered fruit flies – rather than in tissue culture. To locate and quantify the activity of the different caspases in the flies’ bodies under a microscope, the scientists first inserted “reporter” genes into the flies’ genomes, and then relied on these reporters to highlight the proteins to be destroyed with a fluorescent dye.
By showing that the function of caspases is far more complex than previously thought, the new Weizmann study may have important implications for cancer drug development. In particular, the study suggests that in designing drugs intended to enhance apoptosis, researchers must make sure these raise the activity of the caspases above a critical threshold so that cancerous cells are effectively destroyed.
Dr. Eli Arama's research is supported by the Fritz Thyssen Stiftung; the M.D. Moross Institute for Cancer Research; the Y. Leon Benoziyo Institute for Molecular Medicine; the Jeanne and Joseph Nissim Foundation for Life Sciences Research; and the Yeda-Sela Center for Basic Research. Dr. Arama is the incumbent of the Corinne S. Koshland Career Development Chair in Perpetuity.