What scientists didn't know was what regulates the closing of these channels, preventing the cellular warehouses from overfilling and spilling out into the cell. Now, Reuveny, together with Raz Palty, Adi Raveh, Ido Kaminsky and Ruth Meller, have identified
a new protein that helps regulate SOCE activity, as they reported in the journal
Cell. They found that once the Orai channels have opened, the new protein, SARAF, is employed to slowly inactivate STIM, causing the channels to start closing. This prevents the rapid overfilling of cells with calcium and keeps levels under control.
As expected, disabling SARAF activity led to calcium overspill and cell hyperactivity. The added observation that SARAF travels with STIM to the plasma membrane, where the Orai channels are located, provides further evidence that SARAF is involved in regulating STIM deactivation.
Although there is no direct evidence linking mutations in SARAF to human diseases, studies have recently identified SARAF as a biomarker that is associated with prostate cancer, Alzheimer’s disease and dilated cardiomyopathy – disease states that are accompanied by abnormal intracellular calcium levels.
Reuveny: “SARAF is expressed in cells all over the body, but their levels are especially high in the immune system and brain. We still don’t know exactly what it does there or how it works, so this is what we are endeavoring to find out next.”
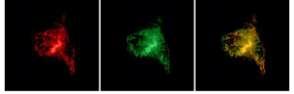
SARAF tagged with green fluorescent protein. The fluorescent signals get stronger as SARAF moves closer to the plasma membrane.
Prof. Eitan Reuveny’s research is supported by the Nella and Leon Benoziyo Center for Neurological Diseases; the Yeda-Sela Center for Basic Research; and the Hugo and Valerie Ramniceanu Foundation. Prof. Reuveny is the incumbent of the Charles H. Hollenberg Professorial Chair.

