In the late nineteenth century, physicians started treating human disease with blood serum extracted from immunized horses. But those extracts – which contained antibodies that target disease agents – often produced disastrous immunological responses. In the late twentieth century, researchers picked up the challenge once again, painstakingly “humanizing” animal antibodies to prevent a violent response from the patient’s immune system. However, replacing an antibody’s animal segments with human ones is laborious, time-consuming and costly; not only that, it can make an antibody less effective or even entirely useless. Now, in a study published in Nature Biomedical Engineering, researchers at the Weizmann Institute of Science have developed an algorithm that works on a new principle, offering a much faster and cheaper way of adapting animal antibodies to make them safe for humans.
Antibodies are proteins that come in countless versions, each capable of neutralizing a specific pathogen after binding to it like a key in a lock. These proteins are Y-shaped molecules, whose upper section consists of two outstretched “hands,” pointing in opposing directions. The hands come with “fingers” that are incredibly variable, which allows them to recognize and grab onto a virtually endless array of pathogens, whereas the Y’s stem region is fairly constant from one antibody to another. Even though an animal antibody often has the right “fingers” to bind to a human pathogen, the rest of the animal molecule might be identified by the human immune system as a foreign invader that must be vanquished. This is why injecting animal antibodies into patients often leads to catastrophe.

“Humanizing an antibody so it can fight disease in our bodies without provoking an immune response can be tricky,” says team leader Prof. Sarel Fleishman of Weizmann’s Biomolecular Sciences Department. He explains that the standard approach has been to select a human antibody that most closely resembles the animal version, then replace the animal segments with human ones while retaining the bits that act against disease. But because different parts of the antibody are interconnected, these replacements can make the antibody unstable and ineffective. “It’s like altering the internal walls of a house without taking its foundations into account,” explains graduate student and lead author Ariel Tennenhouse.
Similarity out, stability in
Fleishman’s lab took up the challenge of coming up with a better approach. The lab is a protein design center, whose members develop computer programs that improve proteins or generate new ones. Since it’s been known for decades that humanizing animal antibodies based on their similarity to human ones often results in unstable molecules, Tennenhouse hypothesized that stability might be a more reliable criterion for humanization. To test this, he developed the CUMAb, an algorithm for computational humanization of antibodies.
Together with collaborators, he then applied CUMAb to explore every possible way to humanize a mouse antibody. The algorithm computed a whopping 20,000 humanized variants of a single antibody and predicted the structural stability of each, selecting the best ones for testing.
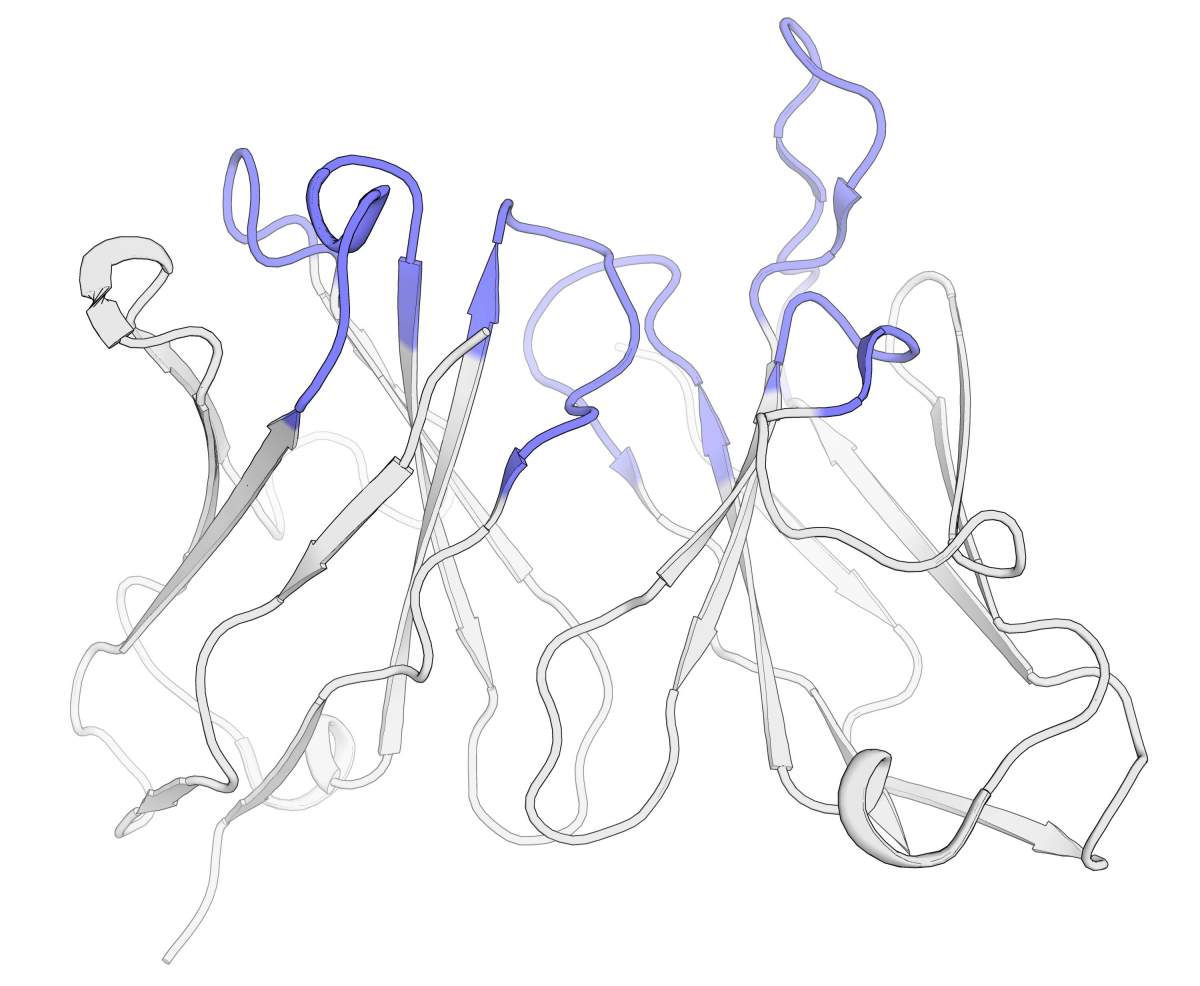
The next step was to create “hard copy” versions of the top-ranking antibodies and test them to see if CUMAb and the hypothesis behind it were correct. The team, together with several other labs, synthesized five different antibodies and tested them experimentally. One of those antibodies was created from a mouse version of an antibody that can stop the growth and spread of cancer cells. Previous attempts to humanize this antibody had come to a dead end. The scientists fed the sequence of the mouse version of this antibody into the program, choosing the most-likely-to-succeed variants to synthesize and test.
""This is the first time that a method has shown such broad success in this critical biomedical engineering problem. It is quite likely to become a key element in accelerating the transition from therapeutic candidate molecules to real-world drugs"
“The results were extraordinary,” says Fleishman. “Without any additional adjustments to the proposed designs, the humanized antibodies functioned just as effectively as the mouse’s original.” Tests of four other antibodies, all designed with CUMAb, were just as impressive, and sometimes even surpassed the original animal antibody’s activity or stability.
CUMAb is a powerful new tool that could significantly accelerate the design of new antibody-based drugs, as well as lower development costs. Fleishman and his team have turned the algorithm into a web server that any academic can use. “This is the first time that a method has shown such broad success in this critical biomedical engineering problem. It is quite likely to become a key element in accelerating the transition from therapeutic candidate molecules to real-world drugs,” says Fleishman.
The new approach to humanizing antibodies might be applied to other issues in therapeutic antibody engineering. For example, many antibody formulations tend to be highly viscous, and they have to be diluted before use. These are then administered through infusion, and it can take hours to get enough of the antibody into the bloodstream. With a computer program that yields a number of viable antibodies, researchers might be able to choose some that require less dilution or have other properties that make them easier or more comfortable to administer, improving outcomes for patients.
“We hope that CUMAb, now accessible online, will empower researchers and professionals to develop antibodies more swiftly and accurately,” says Tennenhouse.