The survival of a species depends in large part on the organisms’ ability to adapt to stressful environmental challenges – for instance, changes in temperature, variations in food supply or pollution. Such adaptations develop over evolutionary timescales as a result of repeated exposures to the environment. But what about rare or extreme events, in which there is not enough time to evolve an adaptive response?
Evidence suggests that certain changes in development can be inherited through so-called epigenetic mechanisms – influences on gene regulation that do not involve a change in the host genome and therefore would not require evolutionary timescales. Do such epigenetic changes – ones that result from short-term environmental pressures – persist across multiple generations? This question, says
Dr. Yoav Soen of the Institute’s Biological Chemistry Department, is rarely addressed.
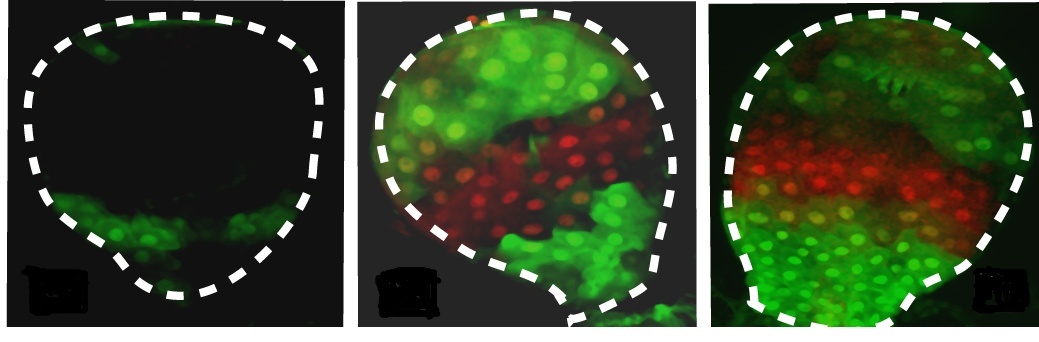
Soen, together with Ph.D. student Shay Stern and Dr. Yael Fridmann Sirkis of the Biological Chemistry Department, and Dr. Erez Braun of the Technion-Israel Institute of Technology, devised a strategy to investigate these issues. To model exposure to an unforeseen environmental challenge, they exposed developing fruit flies to arbitrarily determined patterns of harmful stress across the flies’ tissues, supplementing their food with a toxic drug not normally encountered by flies. The researchers engineered these flies to have a resistance (‘antidote’) gene that was controlled by particular choices of developmental promoters. (Promoters are pieces of DNA that regulate the activation of specific genes.) Because these promoters had not evolved to deal with the drug, there was no effective program already in place for coping with the challenge. Thus the scientists could test how the flies’ systems would handle the situation and whether this could influence the development of their offspring.
The results, recently published in
Cell Reports, show that the survival of flies equipped with the resistance gene depended on how well the promoter controlled that gene (up to 100% survival for one of the promoters). The scientists were also able to uncover part of the mechanism leading to the flies’ survival: Exposure to the toxin repressed a group of genes called Polycomb, whose job is to prevent developmental promoters from being activated in the “wrong” tissues. This repression of the Polycomb system “unlocked” its hold on the promoters, which were then activated outside of their usual tissues. This, in turn, modified the development of the organism. Part of this response was indeed inherited by the fruit flies' offspring, who displayed similar developmental changes even though they were not exposed to the toxic drug. Unlike evolutionary changes, however, these epigenetic modifications were not stable; successive offspring reverted to the species’ original developmental characteristics after several generations.
Further experiments – in which different types of stressors are applied to different stages of development – would be needed to determine the scope and extent of this type of heritable epigenetic change. If the environment can induce epigenetic changes that are stable enough, this might enable diversification, thus influencing the evolution of gene regulation.
Dr. Yoav Soen’s research is supported by the Leona M. and Harry B. Helmsley Charitable Trust; the Y. Leon Benoziyo Institute for Molecular Medicine; and the Yeda-Sela Center for Basic Research. Dr. Soen is the incumbent of the Daniel E. Koshland Sr. Career Development Chair.