One of the mysteries surrounding HIV infection is how the virus manages to hide out in the very immune system cells that are meant to protect the body from harmful invaders. How does the virus prevent these cells from mounting a full-scale attack? In the 22 years since the virus that causes AIDS was identified, much progress has been made in documenting how it breaks into the host cell and reproduces once inside the cell; but many questions remain to be answered. A team of Weizmann Institute scientists has added another piece to the puzzle by showing how a part of a protein on the virus’ outer surface interferes with the cells’ normal immune response. But their work may have wider implications: This molecular fragment, which has such a devastating effect in the case of AIDS, might turn out to be an effective treatment for some chronic disorders such as rheumatoid arthritis. The results of this study were recently published in the Journal of Clinical Investigation.
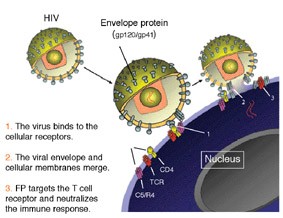
HIV infection begins when the protein coatings of viruses fuse with the outer membranes of T cells - members of the primary response team of the immune system. T cells recognize the presence of foreign invaders and alert other types of immune cells to come to the rescue. The genetic material of the virus, which is basically a strand of RNA, forces the cell’s DNA to make copies of it, and the newly minted viruses created by the host DNA later break out of the cell membrane and infect other cells. Many scientists believed that the very act of breaking into T cells and hijacking their DNA was enough to destroy the ability of these cells to call up immune system reinforcements. But Institute scientists Prof. Yechiel Shai of the Biological Chemistry Department, Prof. Irun Cohen of the Immunology Department and graduate students Francisco Quintana and Doron Gerber thought there must be more to the story. T cells identify invaders using receptors, like security antennae, on their outer walls. Viruses, as well, have their own surface equipment for seeking out specific T cells. To the scientists it seemed unlikely that such a virus would be invisible to the T cell - able to slip past its ever-watchful receptors without raising the alarm. They surmised that the virus must actively disable some part of the immune cell’s system.
They investigated a protein fragment called FP (fusion peptide), which is an active segment of an HIV protein called gp41 found on the outer envelope of the virus. FP was known to play a role in the complex process by which the viral envelope fuses with the cell membrane in the initial stage of cell infection. The researchers suspected that FP, though it is exposed for only a short period during this process, might have enough time, as well as the capability, to deactivate some of the cell’s intruderalert apparatus on its way in. Indeed, they found that FP locks on to several proteins on the cell walls that are involved in invoking a large-scale immune response, effectively shutting them down.
Armed with their new understanding of how a tiny virus can gain control of the body’s immune response, the scientists made an intuitive leap. Could FP, on its own, be an effective tool for controlling harmful immune system activity?
In autoimmune diseases, the same T cells that play host to HIV are overactive, mistakenly attacking the body’s cells instead of foreign invaders. The researchers theorized that, since FP selectively blocks one type of immune response, maybe it could be used in a beneficial manner to override the cells’ misguided call for help in cases of autoimmune disease. To check this theory, the research team tested FP on rats suffering from an autoimmune syndrome similar to human rheumatoid arthritis. As they predicted, the rats treated with FP showed a significant reduction in joint swelling and other symptoms of arthritis.
Shai points out that using FP, a tiny piece of HIV, would pose no danger to patients as it lacks the ability to either infect cells or reproduce. Indeed, as the scientists note in their paper, the study of a destructive virus may contain important lessons for medical science on how to regulate the immune system. “Perhaps,” says Cohen, “we can adopt the virus peptide to help control overactive autoimmunity.”
Prof. Yechiel Shai’s research is supported by the Robert Koch Minerva Center for Research in Autoimmune Disease and the Estate of Julius and Hanna Rosen. Prof. Shai is the incumbent of the Harold S. and Harriet B. Brady Professorial Chair in Cancer Research.
Prof. Irun Cohen’s research is supported by Minna James Heineman Stiftung; the Robert Koch Minerva Center for Research in Autoimmune Disease; and Mr. and Mrs. Samuel Theodore Cohen. Prof. Cohen is the incumbent of the Helen and Morris Mauerberger Professorial Chair in Immunology.