Are you a journalist? Please sign up here for our press releases
Subscribe to our monthly newsletter:

Prof. Lea Eisenbach of the Weizmann Institute’s Immunology Department wants to persuade the body’s immune system to fight cancer. The field of cancer immunotherapy – making use of the immune system’s weapons against malignancy – was selected the "Breakthrough of the Year" for 2013 by Science magazine. But much research is still needed to make these therapies safer and more effective.
The idea for immune-based therapy arose from the fact that the immune system constantly clears away cells that have become cancerous or are on their way to turn malignant. But obviously, since many people do get the disease, some cancer cells manage to evade the immune system’s defenses. Eisenbach is devising several strategies – so far tested only in laboratory animals – to outwit those cancer cells, enabling the immune system to seek out and destroy them.
As a first step, she and her team identified a number of proteins and protein fragments that in cancerous tumors are mutated or present in higher than normal amounts. In experiments in mice, these protein fragments were used as a vaccine that eliminated lung cancer metastases and other tumors, including those produced in mice by various human cancer cells.
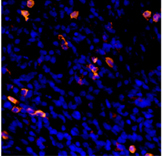
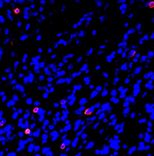
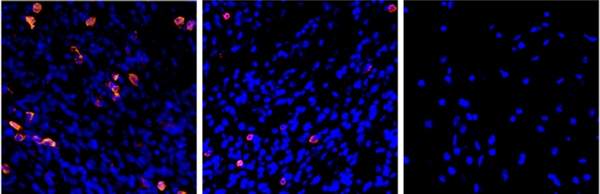
To further develop anti-cancer vaccines, Eisenbach’s team and their collaborators in the MIGAL Research Institute in Kiryat Shmona are teaching the immune system to detect evasive cancers with the help of dendritic cells, the system’s sentinels that normally alert it to the presence of viruses, bacteria and other dangers. The scientists achieve this goal by outfitting dendritic cells with the above-mentioned protein fragments fused with additional molecules that trigger an immune response. By making use of RNA molecules, which convey genetic information but are easier to manipulate than the genes themselves, they have been able to engineer several of the tumor’s genetic features into a dendritic cell, thus increasing the chance that the tumor will be detected.
The engineered dendritic cells can then serve as a vaccine: They migrate to lymph nodes, where they activate the immune system against cancer, priming it into attacking a tumor it previously did not recognize. In a recent review in the Annals of the New York Academy of Sciences, the scientists noted that studies in mice point to the potential efficacy of this approach against metastases of melanoma, particularly those that move to the lungs.
Prof. Lea Eisenbach's research is supported by the Jeanne and Joseph Nissim Foundation for Life Sciences Research; the Victor Pastor Fund for Cellular Disease Research; the Pearl Welinsky Merlo Foundation Scientific Progress Research Fund; the Lewis Family Charitable Trust; and the estate of John Hunter. Prof. Eisenbach is the incumbent of the Georg F. Duckwitz Professorial Chair of Cancer Research.