A murderer turns out to be a saver of lives – this sounds like a plot twist in a thriller, but Weizmann Institute scientists have found that such a scenario can play out in cancer.
In a study published recently in
Cancer Research, they have discovered that a cancer-causing gene can prevent the deadly spread of a tumor at a later point in the progress of the malignancy.
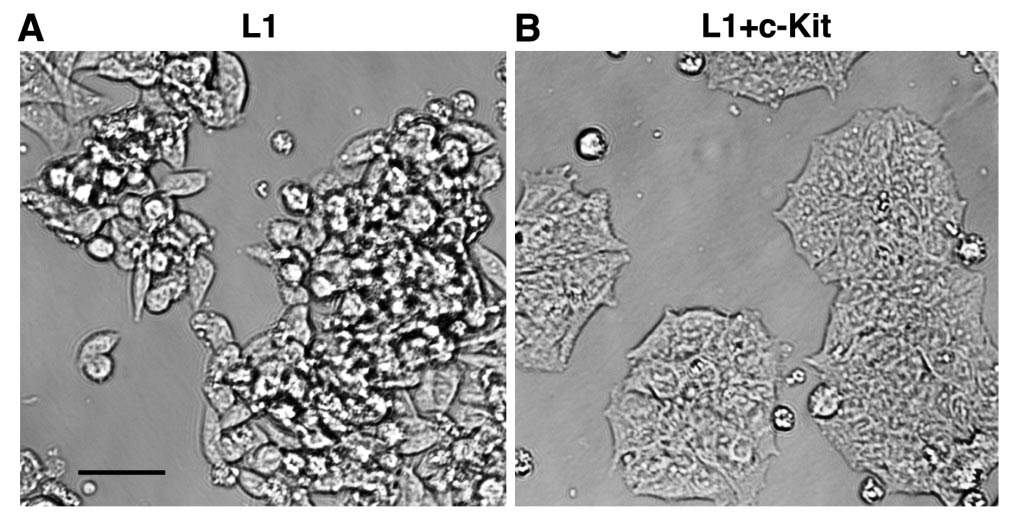
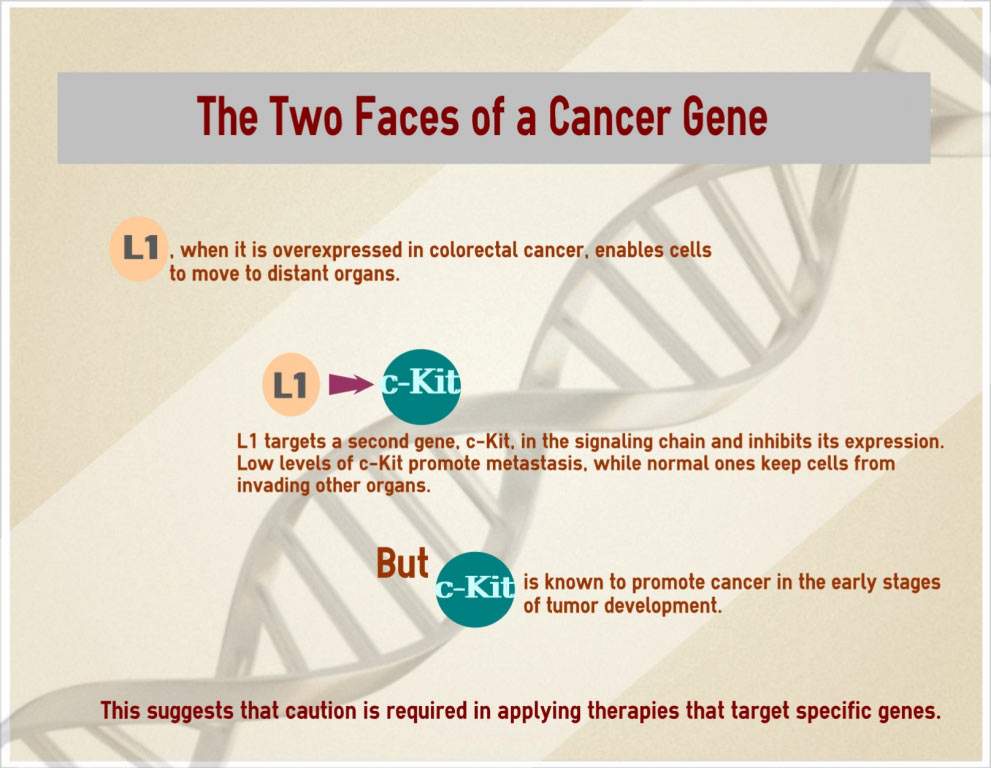
The study’s goal was to identify genes involved in the metastatic spread of colorectal cancer. Working with mice injected with human colorectal cancer cells, the scientists unraveled a long chain of biochemical events that results in metastasis. This chain starts with a mutation present in over 80 percent of patients with malignant colorectal tumors that disrupts a cellular signaling mechanism. The scientists found that the disruption, in turn, increases the expression of a cellular adhesion gene called L1, which enhances the motility of cells at the invasive front of the tumor, facilitating their metastasis to distant organs, in particular the liver.
Moreover, the scientists discovered that L1 affects the expression of one of the targets in its signaling chain: a gene called c-Kit, which is essential for preventing metastasis. When the levels of expression of this gene were low, the tumors metastasized to the liver. But when normal c-Kit levels were restored, the metastasis stopped even in the presence of L1. The c-Kit gene prevents metastasis by prodding cells to stick together and behave in an orderly fashion, the way they do in normal tissue lining, rather than exhibiting the aggressive, “individualistic” properties of invasive cancer cells.
This finding was a major surprise: c-Kit is known to be an oncogene – that is, a cancer-causing gene. In other words, the same gene that promotes growth in the early stages of tumor development, including the growth of colon cancer cells, has been found to block their spread at later stages!
Further analysis suggested that the findings from the mouse study were relevant to humans. When the scientists examined the genomic profiles of tumors in 300 patients with colorectal cancer, they found that in all these patients, c-Kit’s levels were suppressed compared to its levels in the healthy colon.
The research team, headed by
Prof. Avri Ben-Ze’ev of the Molecular Cell Biology Department, included Dr. Nancy Gavert, Anna Shvab, Dr. Amir Ben-Shmuel, Gal Haase and Eszter Bakos. The research was performed in collaboration with Dr. Michal Sheffer from the computational group of
Prof. Eytan Domany of the Physics of Complex Systems Department.
In addition to revealing one of the molecular mechanisms by which colorectal tumors spread to the liver, the most common site of their metastasis, the study has exposed cancer in all its complexity: The same gene can be a “bad guy” at one stage of malignancy and a “good guy” at another. Weizmann Institute researchers conclude that great caution is required in cancer therapies targeting specific oncogenes. In particular, if drugs are given to block c-Kit so as to stop the growth of colorectal cancer, care must be taken not to interfere with the function of this same gene in preventing metastasis. This conclusion is reinforced by recent studies conducted by other researchers. They had shown that another common oncogene, c-Myc, displays the same duality as c-Kit: while it enhances the growth of breast cancer cells, when overexpressed it prevents their metastatic spread.
Prof. Avri Ben-Ze'ev is the incumbent of the Samuel Lunenfeld-Reuben Kunin Chair of Genetics.
Prof. Eytan Domany's research is supported by the Kahn Family Research Center for Systems Biology of the Human Cell, which he heads; the Mario Negri Institute for Pharmacological Research -Weizmann Institute of Science Exchange Program; the Leir Charitable Foundations; Mordechai Segal, Israel; and the Louis and Fannie Tolz Collaborative Research Project. Prof. Domany is the incumbent of the Henry J. Leir Professorial Chair.