The Weizmann Institute was the first academic institution in Israel to create a technology transfer organization for promoting the commercialization of its research.
Yeda Research and Development Co. Ltd., founded in 1959, is charged with finding the right industrial partners to take Institute discoveries through the critical steps from the laboratory to the marketplace.
Scores of patents are granted to Yeda each year for everything from genetically engineered organisms to new materials, software, and medications.
Yeda licenses these internationally, often in partnership with firms based in the nearby Kiryat Weizmann Industrial Park. This high-tech industrial park, Israel's first, was established at the initiative of the Weizmann Institute. Its success gave rise to a second local park, which is now undergoing development.
- Heavy oxygen for the world
- Through a glass darkly
- Diamonds cut - by laser
- Lasers that control chemical reactions
- A weapon to fight malignancies
- A way to rehabilitate the immune system
- Treatment for bone weakness
- Halting osteoporosis
- Two drugs to treat multiple sclerosis
- A versatile peptide
- Improved vaccine against hepatitis B virus
- Where nerves meet muscles (and other nerves)
- New hope for persons at risk of developing diabetes
- Inflammation heal thyself
- Slow release for chronic disease treatment
- Combating bilharzia
- Three-pronged attack on dysentery
- Drainage takes the pressure off glaucoma
- Protection of nerve fibers in glaucoma
- The value of mother's milk
- Viruses under arrest
- Jewish hereditary diseases
- How cells respond to insulin
- The links between diabetes and obesity
- The cloning of SOD
Going to Market
Heavy oxygen for the world
In the 1950s, Weizmann Institute scientists planned and constructed a unique apparatus to separate isotopes. Different isotopes of the same element contain an identical number of protons and electrons, but differ in the number of neutrons in their nucleus, making them heavier or lighter. The Weizmann isotope separator was used to produce isotopes of heavy oxygen: Oxygen-17 (which makes up 0.037 percent of natural oxygen) and Oxygen-18 (0.204 percent of natural oxygen). These isotopes are primarily used for research in chemistry, physics, biology, and medicine.
Their uses include studying nutritional problems in young children and pregnant women, and testing energy consumption in muscles. Scientists also use them in studying aspects of neurological diseases of the brain such as Parkinson's and Alzheimer's. Most of the apparatus' production was for export and for many years it supplied most of the world's demand for heavy oxygen.
Through a glass darkly
Sunglasses and vehicle windshields which darken when bright light falls on them are familiar consumer goods that were developed following the discovery of photochromism, a reversible change in the color of materials due to exposure to light. This discovery was carried out in one of the Weizmann Institute's laboratories. An additional development was recently recorded in this field: a very active photochromic polymer that may be used for the production of photochromic plastic lenses, providing an improved and lightweight alternative to light-sensitive glass lenses.
Diamonds cut - by laser
Weizmann Institute scientists developed an advanced method for cutting diamonds with lasers. This should bring about automation of the diamond industry and introduce ways to design diamonds into unconventional shapes.
Until now the use of lasers for cutting diamonds involved the loss of about five percent of the diamond's weight. The new method practically halved this loss, greatly improving the economic feasibility of using lasers, plus providing other advantages such as better control during the design process.
Lasers that control chemical reactions
Weizmann Institute scientists, together with colleagues from the University of Toronto, developed a technique that uses lasers to control chemical reactions. For the pharmaceutical industry, this method, known as "coherent control," could lead to a new way of separating out biologically-active molecules from their inactive mirror images. In microelectronics, these findings may give rise to much faster switches, operating within a millionth of a millionth of a second. In optics, "coherent control" could bring about a technology for projecting images in mid-air.
New Drugs and Treatment Methods
A weapon to fight malignancies
The protein interleukin-6 serves the body as a sort of messenger that activates the immune system against specific foreign invaders. Interleukin-6 was discovered at the Weizmann Institute, as was the gene responsible for its production and means of producing large quantities of it using genetic engineering techniques.
Interleukin-6 increases the production of blood cells. It also stimulates the body's immune defenses against specific types of malignant tumors that create an abundance of new cancerous growth, known as metastases. This protein and the receptor that augments its activity are produced in Israel and other countries for a variety of medical treatments.
Weizmann Institute scientists later developed peptides that retard interleukin-6 activity, thereby fighting inflammation and myeloma, a specific type of blood cancer.
A way to rehabilitate the immune system
Weizmann Institute scientists isolated and synthesized the thymus hormone, which is a short peptide called THF gamma 2. The hormone works in the human immune system, where it plays an important role in rehabilitating the system, for example after viral infection, chemotherapy, or radiation treatment.
Treatment for bone weakness
Two synthetic derivatives of vitamin D that were developed at the Weizmann Institute are currently being produced on a commercial scale for treating bone weakness associated with kidney disease. One of these products is used as a veterinary preparation against milk fever, a fatal cow disease following bovine pregnancy. It was also found that derivatives of this type of vitamin D decrease the severity of osteoporosis (calcium depletion in the bone), which is a major cause of fractures of the pelvis and other bones in the elderly, especially among women.
Halting osteoporosis
Weizmann Institute scientists uncovered a connection between menopause and the development of osteoporosis (calcium depletion), which occurs mainly in the bones of elderly women, affecting one out of every two women over the age of 50.
The researchers linked the development of the disease to an early decrease in the level of calcitriol, a substance found in the bones. This substance is an active derivative of vitamin D. It is produced in the kidneys and plays a central role in regulating calcium absorption and bone-building processes. In further studies carried out in cooperation with physicians, it was shown that treatment of osteoporosis by a synthetic derivative that converts to calcitriol in the body, helps to slow the disease without side effects.
Two drugs to treat multiple sclerosis
MS is an autoimmune disease affecting the central nervous system that can cause partial or total paralysis. About one million people around the world currently suffer from this incurable disease which is caused when the body's immune system starts attacking the myelin sheath that surrounds and protects the nerve fibers. Two leading modern treatment methods for MS were developed at the Weizmann Institute of Science.
1. Copolymer 1 (known commercially as Copaxone)
A drug which prevents the "fatal attraction" between immune cells and components of the myelin sheath and induces suppression of the autoimmune neurological damage. The drug was approved by the FDA in the United States and in many other countries after it was successfully tested in clinical trials in the United States and Israel. Results demonstrated that it has almost no adverse side effects and that it decreases the number and severity of recurrent attacks, especially among patients in the primary stages of the disease. Copaxone is produced and marketed by Teva Pharmaceuticals Ltd., which received an exclusive license for it from Yeda Research and Development Co Ltd., the Weizmann Institute's technology transfer operation.
The principle upon which Copolymer 1 is based is currently being applied to the study of other autoimmune diseases such as systemic lupus erythematosus and myasthenia gravis.
2. Interferon-beta
A genetic engineering technique developed by Weizmann Institute scientists is being used for the industrial production of the protein, interferon-beta, in animal cell cultures. In clinical trials it was demonstrated that the protein stops the immune system from attacking nerve cells and in so doing, slows the progress of diseases such as multiple sclerosis. Interferon-beta has also been found to have an application in virology and oncology.
Interferon-beta derived from the Weizmann Institute's process causes fewer side effects than those resulting from interferon-beta produced in bacteria. This is because interferon-beta that has been produced in animal cell cultures is closer to that produced naturally by the human body.
A versatile peptide
Weizmann Institute scientists altered the chemical composition of a natural peptide (a sort of short protein) called VIP (Vasoactive Intestinal Peptide- a peptide that expands blood vessels in the intestine). The medical use of this peptide is currently being examined worldwide. So far, three types of potential treatments have been identified:
* The "improved" VIP is capable of penetrating the barrier that separates the blood system and the brain; therefore, it may be used to treat Alzheimer's disease and certain types of mental retardation;
* The improved VIP may be used in local treatment of male impotence;
* New derivatives of the improved VIP may be used in anticancer treatments.
Improved vaccine against hepatitis B virus
People infected with the hepatitis B virus can, with time, develop cancer of the liver. Weizmann Institute scientists developed, by genetic engineering techniques, an anti-hepatitis B vaccine which provides better protection than other vaccines and should significantly decrease the prevalence of cancer of the liver in countries like China where hepatitis B is common.
Where nerves meet muscles (and other nerves)
Weizmann Institute researchers discovered the molecular spatial structure of the enzyme acetylcholinesterase (AChE). The enzyme breaks down acetylcholine, a chemical known as a neurotransmitter, which transmits impulses from one nerve to another and from nerves to muscles.
The discovery, made by use of X-ray crystallography, may provide a basis for planning new and more effective drugs against Alzheimer's disease, in which there is a deficit of acetylcholine. Inhibition of the action of acetylcholinesterase should help Alzheimer's sufferers. Indeed, the discovery has already enabled scientists to understand the functioning of the drug tacrine, currently used in the treatment of Alzheimer's disease.
This discovery should also assist in the development of drugs that target nerve-muscle (neuromuscular) binding sites. These could, for example, treat people poisoned by insecticides or exposed to a nerve gas.
When the brain orders specific muscles to contract, the order is expressed by the secretion of a neurotransmitter called acetylcholine; it travels from the nerve ending into the junction between the nerve and the muscle. This substance causes muscle contraction. An excessive amount of the neurotransmitter may cause the muscle to become paralyzed in a contracted state . When this happens in the heart or diaphragm muscles, it can cause death. To prevent this happening, AChE is active at the junction, ready to break down any excess of neurotransmitter, relaxing the muscle and enabling it to receive additional instructions from the brain.
Certain snake venom neurotoxins, insecticides, nerve agents and drugs bind to AChE in different ways, but all prevent it from fulfilling its function, thereby causing muscle paralysis and even death. In more recent studies, Weizmann scientists have discovered exactly how the snake venom inhibits the enzyme. Based on their knowledge of the structure of AChE, they were able to ascertain the precise spot on the surface of the enzyme molecule to which the snake venom toxin attaches.
New hope for persons at risk of developing diabetes
Juvenile diabetes (also known as type 1 or insulin-dependent) is an autoimmune disease in which some T cells in the body's immune system mistakenly attack and destroy the cells in the pancreas that would normally produce insulin. Innovative treatments being developed by Weizmann Institute scientists are based on a process using peptides to "re-educate" the disease-causing autoimmune T cells, which are destroying the pancreas cells. These re-educated T cells then inhibit the process which leads to the disease. This approach may help to arrest other autoimmune diseases.
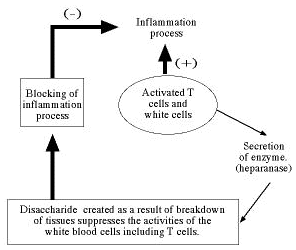
Inflammation heal thyself
Weizmann Institute scientists, together with an Israeli pharmaceutical company, are developing a new method to combat chronic inflammation. Inflammation is characteristic to autoimmune diseases, leading to the rejection of transplanted organs and allergies. The scientists' method is based on using natural by-products of inflammation to inhibit the inflammation process. The method is based on the novel revelation that the immune system itself naturally exploits these small molecules produced by inflammation to regulate the severity of the inflammation. This innovative approach exploits the natural chemical "language" of the immune system for therapeutic purposes.
Slow release for chronic disease treatment
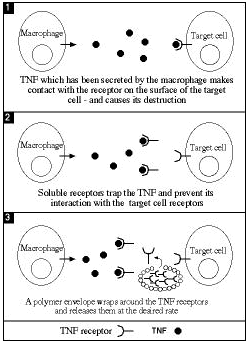
Chronic inflammation involves excessive formation of TNF, a protein that binds to receptors located on the cell surface, causing cell death and an inflammatory reaction. In specific cases, this inflammation causes extreme weight loss (for example, in AIDS patients).
In order to retard the activity of TNF, Weizmann Institute scientists developed a system that allows the use of soluble TNF receptors. These receptors can be injected into the body where they bind to the TNF, thus inhibiting it. However, the body excretes the injected receptors somewhat rapidly, which means that to carry out effective treatment, large quantities are needed. This greatly increases the cost, to the point of impracticality.
To overcome this difficulty, Weizmann Institute researchers, with colleagues from other research institutions, developed a special polymer sheath that contains the TNF receptors and releases them very slowly so that they can inhibit the TNF over a longer period of time. This makes the use of the TNF receptors more effective and economically feasible.
The new polymer sheath should increase the efficacy of many drugs based on receptors of other biological factors which cause numerous and varied diseases. The new treatment has already brought about the cure of rheumatoid arthritis in mice and is currently undergoing clinical trials.
Combating bilharzia
Weizmann Institute scientists isolated immune-reaction stimulating proteins (antigens) of the parasitic worm which causes bilharzia, a debilitating disease that affects millions of people around the world, especially in rural South America, Asia and Africa. These antigens may lead to the development of effective vaccines to combat bilharzia.
Three-pronged attack on dysentery
Weizmann Institute scientists discovered that carriers of the amoebic dysentery parasite, who have no symptoms for many years, may still develop a severe case of dysentery. In light of this, the researchers developed different systems to find the dysentery-causing amoebas in these carriers. These systems are based, among others, on DNA trackers and monoclonal antibodies. The researchers developed advanced techniques to lead drugs to the amoebas to destroy them.
Another study that focuses on antigen proteins on the membrane of the dysentery-causing amoeba is aimed at developing a vaccine against the disease. An additional discovery by Weizmann scientists showed that a specific substance (allicin) found in garlic works potently against the amoebic parasites and other disease-causing microorganisms.
Drainage takes the pressure off glaucoma
In the front part of the eye, where light penetrates, there are no blood vessels. (Blood vessels are important for carrying oxygen and nutrients to cells.) Instead, the cells in this area are nourished by a transparent fluid produced elsewhere in the front of the eye and drained away via a complex drainage system. In glaucoma, this drainage system often malfunctions, causing an excessive accumulation of eye fluid. This increases the pressure inside the eye which can in turn damage the optic nerve.
Weizmann Institute scientists, together with glaucoma specialists from the University of Wisconsin, have proposed a novel approach for increasing fluid drainage, thus decreasing and balancing the pressure inside the eyeball and on the optic nerve. The technique is based on the application of specific drugs for opening new drainage channels between or through the cells that constitute the filter network that drains fluid from the eye. This should augment the drainage rate of the fluid and decrease the pressure it exerts on the optic nerve. The possibility of using this approach to treat glaucoma patients is currently being considered.
Protection of nerve fibers in glaucoma
Weizmann Institute scientists have pioneered the development of an animal model to conduct research on the protection of optic nerve fibers from degeneration, and to examine potential drugs for the treatment of glaucoma. The model is based on the observation that optic nerve fibers damaged by glaucoma continue to degenerate even after the factors causing the disease, such as increased pressure inside the eyeball, have been eliminated. The scientists suggested that the continuing degeneration of the nerve results from the presence of toxic substances derived from the damaged fibers. Inhibiting the activity of these substances, or increasing the resistance of the remaining nerve cells to their toxic effects, may therefore halt the advancement of glaucoma and the progression of blindness. Several synthetic substances tested in the Weizmann model have already been found to have potential as a basis for glaucoma-inhibiting drugs.
The value of mother's milk
Weizmann Institute researchers played a central role in discovering the presence, in breast milk, of peptides that are vital for growth and development. In addition, they discovered that the neurohormone GnRH, responsible for regulation of the reproductive system and for several additional processes occurring in the brain, is produced not only in the brain but also in the breast itself and is secreted with the milk. This finding has important ramifications in understanding the role of the mammary glands in regulating development and sexual functioning.
Viruses under arrest
When a virus infects the body, it invades the cells and forces them to synthesize large quantities of new viruses. Now virologists from the Weizmann Institute have discovered a protein (sLDLR) that is produced by the body in reaction to viral infections, preventing the emission of new viruses from within the body cells. This inhibits the spread of the viral infection, and should be effective in treating various viral diseases, ranging from the flu to AIDS.
Jewish hereditary diseases
Weizmann Institute scientists were among those who assisted in the development of new ways to diagnose and research hereditary diseases that are particularly prevalent in Jewish populations, such as certain types of breast cancer and ovarian cancer, Tay-Sachs disease, a specific type of hemolytic anemia (nicknamed the "Ful" (Broadbean) disease), Gaucher's disease and Niemann-Pick disease.
Tay-Sachs disease, for example, occurs as a result of an accumulation of a specific lipid (ganglioside) in nerve cells in the brain, perturbing their activity. The accumulation results from a deficiency of one form of an enzyme responsible for the breakdown of that particular lipid. Tay-Sachs patients lack this active form of the enzyme and therefore cannot break down these specific gangliosides effectively.
Weizmann Institute scientists developed specific antibodies that bind selectively to each of the enzyme's two versions. This enabled researchers to distinguish and ascertain the quantitative relationship between them. This paved the way for early diagnosis- the ability to identify embryos susceptible to this disease.
Gaucher's disease is similar to Tay-Sachs, but other lipids and enzymes are involved. Weizmann Institute scientists identified some of the genetic mutations responsible for the development of this disease.
How cells respond to insulin
Insulin is a hormone that regulates the amount of sugar in the bloodstream and how much of it reaches the cells, where the sugar is broken down to release energy. Diabetes is caused by a shortage of insulin (known as Type 1, juvenile or insulin-dependent diabetes) or by the cells' failure to respond to the insulin present in the bloodstream (known as Type 2, adult-onset or non-insulin-dependent diabetes).
Currently, many diabetics are treated with injections of insulin directly into the bloodstream. This method provides a solution to those with a shortage of insulin, but does not help when the problem is insulin resistance, a condition in which the cells do not respond to insulin. At the beginning of the process in which insulin affects body tissue, the hormone binds to a specific receptor on the cell membrane. This stimulates a biochemical communication process that enables the appropriate response to insulin. Weizmann Institute scientists identified a specific site in the receptor that is involved in initiating this communication process. They discovered that insulin resistance may occur because some of the proteins that relay messages from the insulin receptor to the glucose uptake machinery in the cell undergo excessive phosphorylation (a process during which they acquire phosphorus atoms). Such abnormal phosphorylation generates a "short circuit" that prevents the insulin receptors from communicating their "messages" to the cells' interior, making the cell resistant to insulin.
In addition, the new study may explain why diabetes is five times more common in obese people than in people of normal weight. A major culprit in enhancing the inappropriate phosphorylation is a molecule called TNF, which is secreted by fat cells. The excessive secretion of TNF by obese people may explain the link between obesity, insulin resistance and diabetes. These findings may in future lead to new treatments for diabetes that would be based on correcting insulin resistance.
The links between diabetes and obesity
It is not known why millions of diabetes sufferers worldwide develop this disease. Particular forms of diabetes are expressed by a weakening of the sensitivity of the body's cells to the hormone insulin, which plays a central role in converting sugars into energy-storing substances such as complex sugars (polysaccharides) and fats. When this process isn't carried out efficiently, the sugars remain in the blood. For many years scientists and researchers all over the world have been trying to understand why the body's cells lose their ability to react to insulin. Two agents affecting insulin action were identified: free fatty acids and TNF-alpha. Now Weizmann Institute scientists have also identified a third agent.
The scientists discovered that a quantitative increase of a certain protein, leptin, interferes with insulin's normal activity. Leptin became famous in 1994 when American researchers injected it into genetically-obese mice and reduced their weight. This result raised hopes of developing a weight-reducing drug based on leptin.
However, overweight people (who have a greater tendency to develop diabetes) already have a relatively large amount of leptin in their blood. (Leptin is produced in fat cells.) Institute scientists have discovered that leptin at concentrations found in obese individuals interferes with some of insulin's activities. This sounded a warning bell: any weight-reducing drugs based on leptin may turn out to be an accelerating factor in the development of diabetes.
The cloning of SOD
Weizmann Institute scientists cloned the gene responsible for the production of the SOD enzyme in humans. They later developed, in cooperation with colleagues in the biotechnology industry, a way to synthesize and produce this enzyme in bacteria in large quantities, using genetic engineering techniques. The enzyme produced by this method may help in the treatment of prematurely-born infants with respiratory difficulties. This is particularly important given the rise in the number of premature births stemming from the increased use of fertility treatments.






















